INFANT FORMULA
Infant formula is considered a specific and unique category of dairy products in China. In recognition that these are products designed for the most vulnerable consumer group, infants, the Chinese regulators have implemented a specific process of establishment registration and subsequently brand registration to ensure the products available in the market are subject to rigorous assessment and review to provide the highest possible level of food safety.
The process for gaining CNCA registration for Infant Formula manufacturers requires completion of a specific set of documentation.
The registration process requires that the completed documentation is provided to the CNCA via DAWR in Australia. Once the documents have been reviewed, the CNCA will then need to undertake a physical audit of the establishment. New exporters wishing to ship infant formula products to China should be aware that this can be a lengthy process. Recent experience has seen several Australian applicants wait for 16 months to have their establishment audited, and three months later are awaiting the report from this audit.
In addition to the CNCA registration process, the CFDA have implemented a requirement for CNCA registered establishments to register their brand formulations.
Brands are able to gain clearance for sale into the China market after 1 January 2018 if they have not been granted CFDA brand registration. Establishments may apply for up to 3 brands to be registered with the CFDA, and each brand must disclose the details of the formulation, and scientific justification for the ingredients used to demonstrate the nutritional value. Each of the three brands must have formulations that are significantly different.
The dossier to be submitted for the brand application is very detailed. A checklist if items to be included can be found in the table below.
- Application form for product formula registration of infants and young children formula milk powder
- Evidentiary documents proving the subject qualification of the applicant
- Quality and safety standards for the raw and auxiliary materials
- Product formulation
- Product formula research and development report
- Explanation of production process
- Product inspection report
- Evidentiary materials of production ability, R&D ability and inspection ability
- Samples of labels and instructions as well as explanatory and evidentiary materials of claims thereof.
It should also be noted that all dairy products exported to China are governed by the requirements of the Food Safety Law. Dairy products must all meet the requirements of the relevant GB standards.
What are GB standards?
GB standards are China’s national standards, also known as ‘Guo Biao Standards’. China’s GB Standards are classified in two stages by their prefix code: “GB” are Mandatory standards; “GB/T” are Recommended standards (Quasi-Mandatory standards).
Mandatory standards have the force of the law as do other technical regulations in China. They are enforced by laws and administrative regulations and concern the protection of human health, personal property and safety.
Recommended standards
In addition to the General Standards which may apply, there are specific GB Product Standards for each product category which must be complied with. Click the menu item below titled “GB Standards” to download the relevant GB Product Standard for this product, as a PDF file.
Use the easy-to-follow checklist (clickable links) below to help you establish all the necessary requirements needed to export Infant Formula into China.
Important additions to Infant Formula
It’s difficult to arrange a time for visits to plants. The CNCA has a small team of approved auditors for this process, and this process applies to all countries, not just Australia. This means it’s unlikely they will visit to audit a single factory.
In some cases, Australian plants have waited up to 18 months to be audited. We now have 15 registered establishments approved for IF exports to China; one of the highest number for any single country in the world
In 2017, the CFDA introduced that infant formula brands must also be registered (not just the plants they are made in). There is also a restriction on the maximum number of brands registered per plant, up to three. There is a high level of scientific data that must be generated and provided to substantiate the recipe – ingredients used, their proportion, and extensive shelf-life validation data. Each of these brands must have unique formulations (that is, you can’t use the same one in three different brand names) and so this is a very complex process.
Market Insights*
When customers are looking for infant formula products, most of them prefer to get information from friends, online communities and offline retail channels, as they prefer to collect non-official comments as much as possible to judge an infant formula product / brand.
Source: 2015 Sina Mother & Baby shopping report, June 2017
Word-of-mouth has significant influence on the decision-making process. When customers are looking for infant formula products, most of them prefer to get information from friends, online communities and offline retail channels.
Food safety is the core concern. The perception of quality is influenced by the country of origin and the brand. Australian dairy ranks very highly.
Most infant formula customers prefer to choose infant formula products sold in metal containers versus paper-based/cardboard containers, or plastic sachets.
Price is not a key concern when choosing infant formula products.
Nutrition and infant formula recipes (ie: components which make up the powder) are core concerns for parents when making the decision on products and brands of milk powder.
Offline shopping channels including maternity stores and supermarkets, play a major role in infant formula purchasing.
e-shopping on major B2C platforms is largely conducted by customers from East China (Shanghai, Zhejiang, Jiangsu, Anhui, Fujian, Shandong, Jiangxi), North China (Beijing, Tianjin, Hebei, Shanxi) and South China (Guangdong, Guanxi, Hainan, Hong Kong, Macou).
Customers living in East China contribute to half of total infant formula shopping.
East China and South China customers prefer to shop through general e-commerce channels such as JD.
* All data published on this website is to be used for indicative purposes only and should not form the basis of major decisions to export dairy products to China. Facts and insights have been sourced from Daxue Consulting Research Report, June 2017. All rights reserved.
Case Example
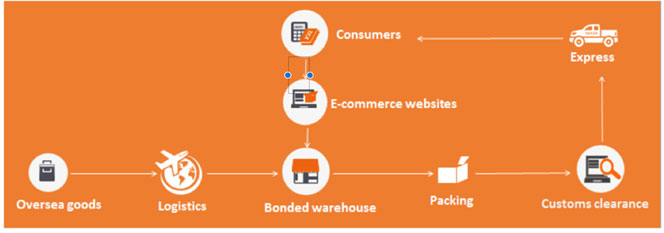
Infant Formula imported through general trade
Before importation
Stage 1: Manufacturer registered by CNCA
First, please confirm the manufacturer has been registered by CNCA through checking whether the name of the manufacturer has been listed in CNCA official website.
Stage 2: Recipe registered by CFDA
Second, the manufacturer shall apply for CFDA recipe registration. Required dossiers shall be prepared in Chinese and must include Chinese product labels. The product test report must be compliant with Administrative Measures of Infant formula recipe registration. This means the test method shall be GB methods. The cost of testing will be high, as all declared elements of the nutrition information panel must be analysed and quantities validated.
Stage 3: Importer and exporter filing
After confirming that the manufacturer has been registered by CNCA and the recipe has been registered by CFDA, the infant formula can be imported to China. Before importation, all of the steps in each of the menu tabs under Infant Formula must be completed, starting at ‘Importer Qualifications’ through to ‘Traceability’.
Importer Qualifications
Chinese importer is the party who, for customs purpose, makes (or on whose behalf an agent or broker makes) the import declaration, and who is liable for the payment of duties (if any) on the imported goods. Normally, this party is named either as the consignee in the shipping documents and/or as the buyer in the exporter’s invoice. The Chinese importer should be registered in China and has the import qualification.
Once the goods arrived at the designated port, the importer will first transfer the goods to the customs supervisory warehouse, and then handle the customs clearance formalities with required documents and materials, including but not limited to import commodity inspection and customs declaration. Since the procedure is very complicated and involves multi-stakeholders, to complete it smoothly, cooperating with an experienced and qualified importer is very critical. After successfully go through all the procedure, the importer will pick up the goods and get it enter the Chinese market.
Customs Inspection Credit Management System
What is Customs Inspection Credit Management System?
Customs Inspection “Credit Management System” refers to the procedure that China Customs Inspection collect and review the credit information of recorded enterprises and then give those enterprises certain credit rate.
The Credit Management System impacts the following enterprises:
- Exporters/Importers (such as Australia exporters/agents of imported foods, Chinese importers);
- Agents for inspection declaration, entry-exit inspection operators and quarantine processing entities in China;
- Port food production and operation entities, storage entities, inspection and identification institutions in China;
- Other inspection and quarantine supervision and administration objects for which credit management is necessary Customs Inspection “Credit Management System” enables the customs inspection to score and evaluate the enterprise’s inspection credibility and violations of any, the higher the credit the better preferential treatment the enterprise gets hence smoother imports in to China.
What is the initial ranking for new enterprises?
Newly established enterprises will be granted Category ‘B’.
What is the ranking evaluation period and methodology?
A ranking for Category ‘A’ to ‘D’ will generally be made based on a one-year rating cycle and according to China Customs Inspection enterprises do not need to apply for Category ‘A’ to ‘D’. The ranking will be granted by the in-charge local Customs Inspection and enterprises can consult with the in-charge Customs Inspection thereafter.
What is the evaluation standard?
The ranking standard is from Category ‘AA’ to Category ‘D’. The initial credit score for each company is 100 and deductions will be made thereafter based on any identified violations. The score range for each Category is tabled below:
| Category | Score | |
| A | 89-100 | |
| B | 77-88 | |
| C | 65-76 | |
| D | 0-64 | |
Dynamic management rules apply and these refer to the immediate measures that Customs Inspection will take against enterprises based on the deduction obtained within one evaluation period. Set out below is a table for deduction points and corresponding management measures:
| Annual Deduced Points | Management Measures | |
| 12-23 | “Stake-out” (i.e. tightening the supervision) | |
| 24-35 | “Immediate downgrading” and “Stake-out” | |
| ?36 | “Included in the list of serious and dishonest enterprises in the list of serious and dishonest enterprises” (i.e. published) with tightened supervision and downgrade to Category “D”. | |
Who is responsible for violations and points deductions therein?
When multiple enterprises are involved, deduction will be determined on a case by case basis in terms of the corresponding enterprise’s responsibility.
What preferential treatment is granted based on the ranking?
Detailed preferential treatment is likely to vary in different regions and local Customs Inspection should submit pilot plans on the preferential treatment for central China Customs review and approval. The preferential treatment will most likely be granted not only based on the ranking of the enterprises but also on the risk of the imported/exported products. In principle, the preferential treatment to be granted by the local in-charge Customs Inspection to the enterprise should be as follows:
| Category | Preferential treatment | |
| AA | Enjoy all preferential treatment as granted to Category “A” Priority for inspection declaration, quarantine and release Priority for appointment of inspection declaration Priority for filing, registration and other procedures Priority for trial of new trade measures |
|
| A | Access to inspection and quarantine incentives Priority for implementation of “class one” preferential inspection, quarantine, green channel and related trade facilitation measures |
|
| B | Carry out daily supervision, inspection declaration, inspection and quarantine, release and other processed in conjunction with the relevant provisions | |
| C | Supervised in a strengthened manner | |
| D | Supervised with restricted administration measures Reassess the obtained qualification |
|
| A Category “A” enterprise can apply for Category “AA” subject to meeting certain additional criterion. | ||
How to know the credit ranking of the enterprise?
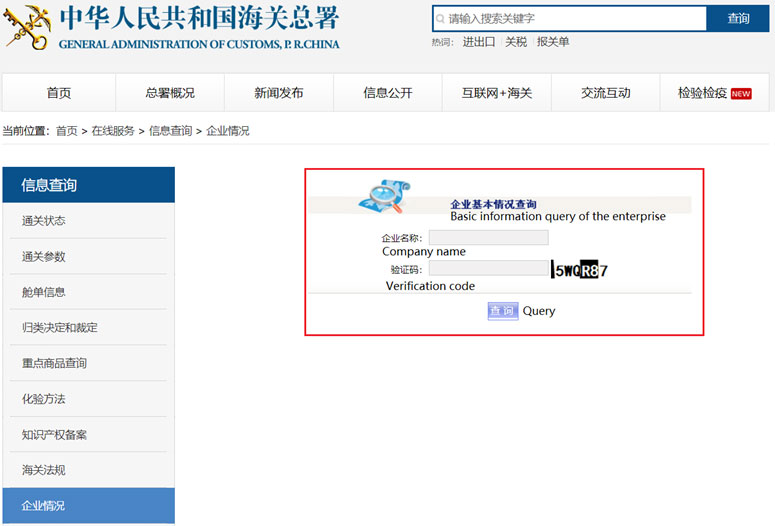
Enterprise may achieve its credit ranking by asking local Customs. Or search from the following official website of China Customs by the Chinese name of the enterprise, the steps are as below.
Step 1 Access to http://www.customs.gov.cn/customs/302427/302442/qyqk/index.html, you will see the following page
Step 2 Within the red square, input the Chinese company name and verification code in the corresponding table. Then click query, the company information will appear below the query table. The credit rating is in the fourth column.
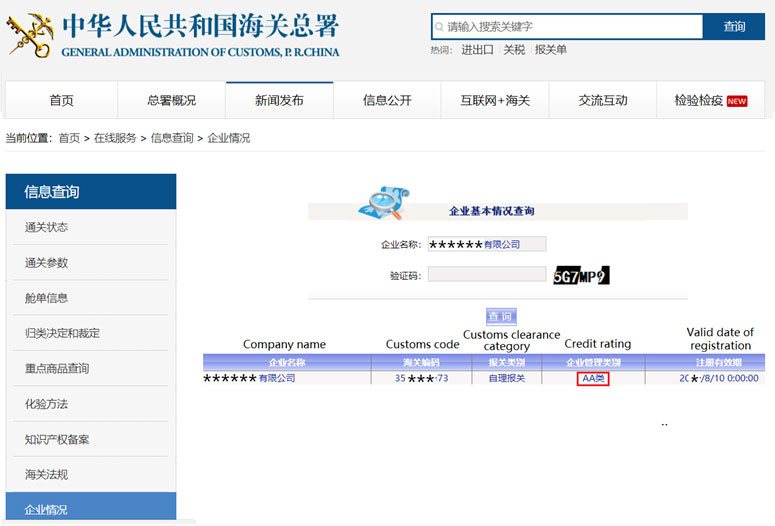
Note: This system can only use the company’s Chinese name to do research. To ensure the accuracy of the results, please use the official Chinese name of the company, which is the company name in the business license. Besides, you may use the customs code in the second column to check.
A list of useful questions to ask to check whether your importer has the qualifications
| No. | Questions | Expected answer | ||
| 1 | Can you show me your business license and food business license? | Yes | ||
| 2 | Do you have the right of importing dairy products? | Yes | ||
| 3 | Have you completed the AQSIQ registration for importers within borders? | Yes | ||
| 4 | Have you recorded in China Chamber of Commerce of Foodstuffs and Native Produce (this question is only for importing dairy products with following HS code: 04011000, 04012000, 04014000, 04015000, 04021000, 04022100, 04022900, 19011010)? | Yes | ||
| 5 | Do you have automatic Import License (this question is only for importing dairy products with following HS code: 0401100000, 0401200000, 0401400000, 0401500000, 0402100000, 0402210000, 0402290000, 1901101000)? | Yes | ||
| 6 | Can you show me your credit ranking? | Sure | ||
Exporter Filing
1. General Administration of Customs, P.R.China (Customs) is responsible for inspection and accreditation of import and export food.
2. According to “Administrative Provisions on Filing of Importers and Exporters of Foods”, food exporters should conduct filing with Customs through an on-line registration system prior to initial export to China.
3. This simple and free procedure requires inputting basic company information and takes around 3 working days for approval.
How to conduct Customs filing?
Step 1 Access to http://ire.eciq.cn/, you will see the page below, the middle one is for overseas exporter filing.
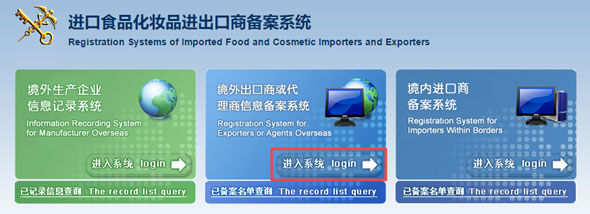
Step 2 Click login, you will be redirected to another page.
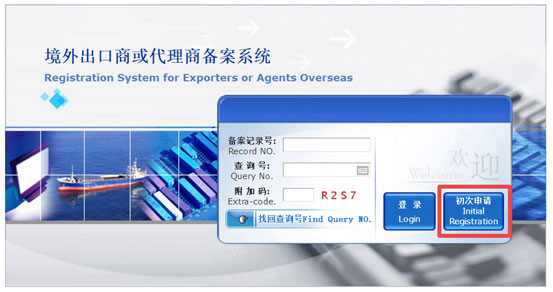
Step 3 “If you have already filed, then you can log in with your Record No. and Query No..
If it is the first time you use this system, click “Initial Registration” and you will be led to the filing page.”
Step 4 “Fill in Applicant’s Information, information marked with an * must be submitted.
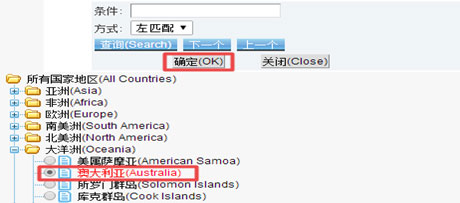
Choose “Exporter” as the company type. For “Country/Area”, click on the question mark, you will be led to a new page, then select “Oceania→Australia” and click “OK”.”
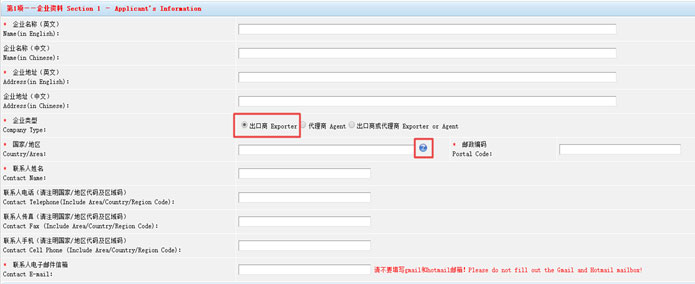
1. Once finished, all information can be edited except for Name (in English), Address (in English) and Country/Area.
2. Please do not fill out the Gmail and Hotmail in the Contact E-mail.
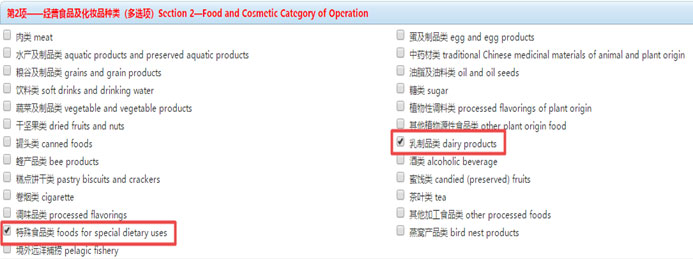
Step 5 Choose the category of “dairy products” and “foods for special dietary uses”.
Step 6 Click “Agree” to sign the letter of commitment.
Step 7 Fill in the information of submitter. Once fill in all the information above, click “Commit” to submit the filing information.
Step 8 When submit the above information, you will see a confirmation box. Click “确定” to complete the submission.
Step 9 After submitting, you will receive a prompt message of applying successfully. Please record the registration number and query number so that you can use them to login the system and check the status of filing.
How to conduct Customs filing?
Step 1 Access to http://ire.eciq.cn/, you will see the page below, the middle one is for overseas exporter filing.

Step 2 Click login, you will be redirected to another page.

Step 3 “If you have already filed, then you can log in with your Record No. and Query No..
If it is the first time you use this system, click “Initial Registration” and you will be led to the filing page.”
Step 4 “Fill in Applicant’s Information, information marked with an * must be submitted.
Choose “Exporter” as the company type. For “Country/Area”, click on the question mark, you will be led to a new page, then select “Oceania→Australia” and click “OK”.”

1. Once finished, all information can be edited except for Name (in English), Address (in English) and Country/Area.
2. Please do not fill out the Gmail and Hotmail in the Contact E-mail.

Step 5 Choose the category of “dairy products” and “foods for special dietary uses”.

Step 6 Click “Agree” to sign the letter of commitment.

Step 7 Fill in the information of submitter. Once fill in all the information above, click “Commit” to submit the filing information.

Step 8 When submit the above information, you will see a confirmation box. Click “确定” to complete the submission.

Step 9 After submitting, you will receive a prompt message of applying successfully. Please record the registration number and query number so that you can use them to login the system and check the status of filing.

CNCA Registration
CNCA is the abbreviation of Certification and Accreditation of the People’s Republic of China which is the subsidiary organisation of State Administration for Market Regulation. CNCA registration refers to overseas manufacturers of meat, aquatic, dairy products and bird’s nest which should be registered as approved establishments with CNCA. For dairy products, the registered objects include processing plants, cold stores and dry stores. CNCA registration is a prerequisite and should be accomplished before the products are exported to China.
Do you need to register with CNCA?
Yes, all establishments manufacturing milk-based infant formula must obtain CNCA registration. A list of the establishments that have been audited, approved and listed by the CNCA is available on the CNCA website.
The term of validity of the CNCA registration of manufacturers is 4 years. If a registered manufacturer of infant milk-based formula needs to renew its registration, it shall submit its application one year prior to the expiration of its registration, and provide relevant information and supporting documents to prove that the company continues to comply with the registration requirements as requested.
Applications-related information and supporting documents will be reviewed by the CNCA. For those manufacturers that meet the registration requirements, their registration will be approved or renewed; for those manufacturers that can’t meet the registration requirements, their registration will be refused or suspended; and for those manufacturers who fail to submit applications to registration renewal after the expiration of the registration, their registrations will be cancelled.
How to register and next steps?
Updates to the CNCA infant formula list are submitted to CNCA by the Department of Agriculture and Water Resources (the department). The DAWR can only recommend amendments and deletions to the CNCA infant formula list. Any new establishments must have had a post-production on-site audit before they will be included on the list sent to CNCA by the DAWR. Infant formula businesses must complete a specific Registration Application Form for their establishments (please refer to the part of ‘Dossiers for application’ for more details). For infant formula products CNCA will complete an audit of your manufacturing facility prior to approval and listing.
Please note if infant formula establishments (that have been listed by China) have a change of name, businesses must submit a new CNCA infant formula application form. Below is the CNCA registration procedure:
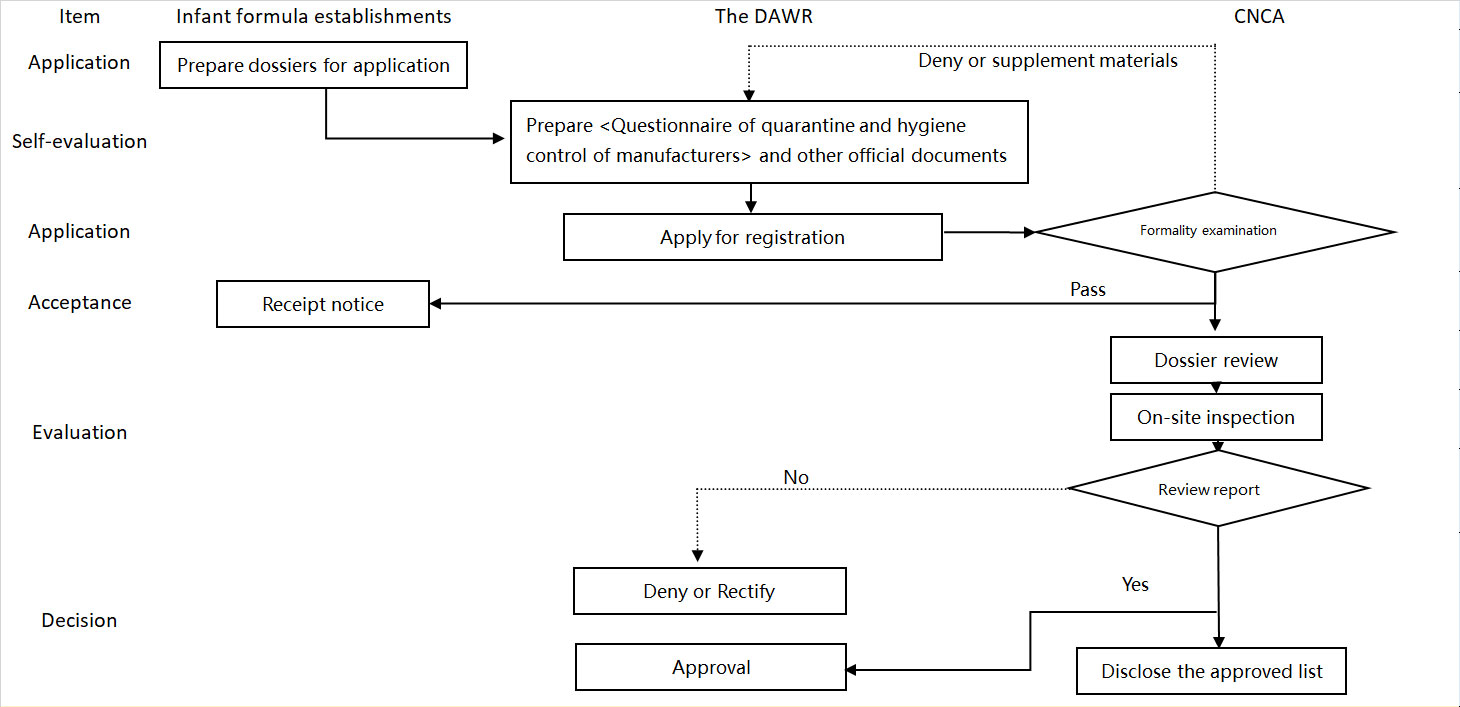
Dossiers for application
The dossiers for application you should prepare mainly refers to the registration application form for infant formula establishments.
The registration application form for infant formula establishments has two parts:
Part I: General Information about the Enterprise – this must include information about production (such as HACCP and CIP), raw materials, product traceability and recall, product testing, and the location and environment of your establishment.
Part II: Overview of the Enterprise’s Exports to China – this must include information about the relationship between the producer, exporter, importer, the trademark holder that exports the products to China and the responsible party that exports the products to China, import information (such as details of the importer, trademarks, consumer complaints system in China and procedures for recalls in China) and export information.
The confirmation form can be paper or electronic, and the language can be either in Chinese or in English. Please find the link of the form below.
English version | Chinese version
Estimated timeline
History would suggest that dairy manufacturers can expect delays of up to 24 months for the CNCA registration of infant formula establishments.
Link to responsible agencies and contacts
To obtain CNCA registration, contact the Department of Agriculture and Water Resources, who will submit the application to CNCA on your behalf.
The contacts are as below:
Tel: 1800 900 090
Website: http://www.agriculture.gov.au/export/controlled-goods/dairy/operating-registered-establishment/china-requirements
Do you need to register with CNCA?
Yes, the establishments manufacturing milk-based older infant formula and young children formula should obtain CNCA registration. A list of the establishments have been audited, approved and listed by the CNCA is available on the CNCA website: http://www.cnca.gov.cn/bsdt/ywzl/jkspjwscpqzc/
The term of validity of the CNCA registration of manufacturers is 4 years. If a registered manufacturer of milk-based older infant formula and young children formula needs to renew its registration, it shall submit its application one year prior to the expiration of its registration, and provide relevant information and supporting documents to prove that the company continuously to comply with the registration requirements as requested.
The application to registration as well as related information and supporting documents, among others will be reviewed by the CNCA. For those manufacturers that meet the registration requirements, their registration will be approved or renewed; for those manufacturers that can’t meet the registration requirements, their registration will be refused or suspended; and for those manufacturers who fail to submit applications to registration renewal after the expiration of the registration, their registrations will be cancelled.
How to register and next steps?
Updates to the CNCA infant formula and young children formula list are submitted to CNCA by the Department of Agriculture and Water Resources (the department). The DAWR can only recommend amendments and deletions to the CNCA infant formula and young children formula list. Any new establishments must have had a post-production on-site audit before they will be included on the list sent to CNCA by the DAWR. Older infant formula and young children formula businesses must complete a specific Registration Application Form for their establishments (please refer to the part of ‘Dossiers for application’ for more details). For milk-based older infant formula and young children formula products CNCA will complete an audit of your manufacturing facility prior to approval and listing. Please note if older infant formula and young children formula establishments (that have been listed by China) have a change of name, businesses must submit a new CNCA application form. Below is the CNCA registration procedure:

Dossiers for application
The dossiers for application you should prepare mainly refers to the registration application form for milk-based older infant formula and young children formula establishments.
The registration application form has two parts:
Part I: General Information about the Enterprise – this must include information about production (such as HACCP and CIP), raw materials, product traceability and recall, product testing, and the location and environment of your establishment.
Part II: Overview of the Enterprise’s Exports to China – this must include information about the relationship among the producer, exporter, importer, the trademark holder that exports the products to China and the responsible party that exports the products to China, import information (such as details of the importer, trademarks, consumer complaints system in China and procedures for recalls in China) and export information.
The application form can be paper or electronic edition, and the language can be either in Chinese or in English. Please find the link of the form below.
English version | Chinese version
Estimated timeline
History would suggest that dairy manufacturers can expect delays of up to 24 months for the CNCA registration of milk-based older infant formula and young children formula.
Link to responsible agencies and contacts
To obtain CNCA registration, please reach out the Department of Agriculture and Water Resources, who will submit the application to CNCA on your behalf.
The contacts are as below:
Email:
Tel: 1800 900 090
Website: http://www.agriculture.gov.au/export/controlled-goods/dairy/operating-registered-establishment/china-requirements
Preparation for product formula
Just like the Australia New Zealand Food Standards Code in Australia, the national food safety standards (GB standards) are counterparts in China. Usually, each category of foods has one corresponding GB product standard, which stipulates the definitions, sensory and physicochemical indexes, limits of contaminants, mycotoxin, pesticides and microbial indicators of the food. The Chinese authority will conduct inspection and quality control based on related GB standards when the products arrive at China ports. If the tested product cannot meet the requirements of GB standards, it will be returned or destroyed.
Abstract of standard
| Application Scope | This standard applies to infant formulas. | |||||
| Terms and definitions | Infants | Refer to persons of 0 ~ 12 months old. | ||||
| Milk-based powder infant formula | Refers to powder products made only through physical methods, of which the main material is milk and milk protein products, supplemented with a proper amount of vitamin, minerals and other supplementary materials, which are applicable to normal infants, where the energy and nutrition can satisfy the requirements of growth and development of normal infants of 0~6 months old. | |||||
| Requirements for Raw Materials | Ingredients and food additives adopted should not contain gluten. | |||||
| Hydrogenated oil and fat should NOT be used. | ||||||
| Raw and supplementary materials treated by irradiation should NOT be used. | ||||||
| For milk-based infant formula, the preferred carbohydrate should be lactose, or lactose and the polymer of glucose. Only after pre-gelatinisation can starch be added into the infant formula. Fructose can NOT be used. | ||||||
| Sensory requirements | Items | Requirements | ||||
| Color | Should conform to the features of related products. | |||||
| Taste and odor | Should conform to the features of related products. | |||||
| Structural state | Should conform to the features of related products. No visible foreign substances should be included in the product. | |||||
| Dissolvability | Should conform to the features of related products. | |||||
| Essential components | All essential components in the product is essential for the growth and development of infants. | |||||
| The energy in the ready-to-eat infant formulas per 100 mL should be within the range of 250 kJ (60 kcal)-295 kJ (70 kcal). The calculation of energy should be the value of the product of the content of protein, fat and carbohydrate per 100 mL product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber should be based on 50% of carbohydrate energy coefficient) respectively, and the sum (kJ/100mL) is divided by 4.184 to kcal/100 mL. | ||||||
| The protein, fat, carbohydrate content of infant formula per 100 kJ (100 kcal) should be consistent with the provisions set out in table below. | ||||||
| Nutrient | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Protein/(g) | 0.45 | 0.70 | 1.88 | 2.93 | For milk based infant formulas, the content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | 1.05 | 1.40 | 4.39 | 5.86 | Among the finished products, the amount of lauric acid and myristic acid (tetradecanoic acid) should account for no more than 20% of the total fatty acid; the maximum content of trans fatty acid should not exceed 3% of the total fatty acid; the erucic acid content should not exceed 1% of the total fatty acid. The total fatty acid refers to the sum of C4~C24 fatty acid. | |
| In which: linoleic acid(g) | 0.07 | 0.33 | 0.29 | 1.38 | ||
| α- linolenic acid (mg) | 12 | N.S. | 50 | N.S. | N.S.: No specification | |
| Linoleic acid/α- linolenic acid ratio | 5:1 | 15:1 | 5:1 | 15:1 | ||
| Total carbohydrate /(g) | ≤5.0 | The content of lactose in total carbohydrate should be over or equal to 90%. For calculation of the proportion of lactose among the total carbohydrates, the added oligosaccharides and polysaccharides should not be included. | ||||
| Carbohydrate content A1 should be calculated according to formula(1): A1= 100 − (A2+ A3+ A4+ A5+ A6) ………………………………(1) Where: A1——carbohydrate content, g/100g; A2——protein content, g/100g; A3——fat content, g/100g; A4——moisture content, g/100g; A5——ash content, g/100g; A6——dietary fiber content, g/100g. |
||||||
| Vitamins | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Vitamin A /(μg RE) | 14 | 43 | 59 | 180 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | 0.25 | 0.60 | 1.05 | 2.51 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | 0.12 | 1.20 | 0.50 | 5.02 | 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3). | |
| Vitamin K1/(μg) | 1.0 | 6.5 | 4.2 | 27.2 | ||
| Vitamin B1/(μg) | 14 | 72 | 59 | 301 | ||
| Vitamin B2/(μg) | 19 | 119 | 80 | 498 | ||
| Vitamin B6/(μg) | 8.5 | 45.0 | 35.6 | 188.3 | ||
| Vitamin B12/(μg) | 0.025 | 0.360 | 0.105 | 1.506 | ||
| Niacin (niacinamide)/(μg) | 70 | 360 | 293 | 1506 | Niacin: excludes precursor form. | |
| Folic acid/(μg) | 2.5 | 12.0 | 10.5 | 50.2 | ||
| Pantothenic acid/(μg) | 96 | 478 | 402 | 2000 | ||
| Vitamin C /(mg) | 2.5 | 17.0 | 10.5 | 71.1 | ||
| Biotin/(μg) | 0.4 | 2.4 | 1.5 | 10.0 | ||
| Minerals | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Sodium/(mg) | 5 | 14 | 21 | 59 | ||
| Potassium/(mg) | 14 | 43 | 59 | 180 | ||
| Copper/(μg) | 8.5 | 29 | 35.6 | 121.3 | ||
| Magnesium/(mg) | 1.2 | 3.6 | 5 | 15.1 | ||
| Iron/(mg) | 0.1 | 0.36 | 0.42 | 1.51 | ||
| Zinc/(mg) | 0.12 | 0.36 | 0.5 | 1.51 | ||
| Manganese/(mg) | 1.2 | 24 | 5 | 100.4 | ||
| Calcium/(mg) | 12 | 35 | 50 | 146 | ||
| Phosphorus/(mg) | 6 | 24 | 25 | 100 | ||
| Calcium/phosphorus ratio | 1:1 | 2:1 | 1:1 | 2:1 | ||
| Iodine/(μg) | 2.5 | 14 | 10.5 | 58.6 | ||
| Chloride/(mg) | 12 | 38 | 50 | 159 | ||
| Selenium/(μg) | 0.48 | 1.9 | 2.01 | 7.95 | ||
| Optional components | In addition to the essential components in the table above, one or more nutrients listed in Table below can be selected to add or claimed on label, whereas the content of such nutrients should meet the specification of Table below. | |||||
| To improve the protein quality of infant formula or enhance its nutritional value, the monomer L-amino acid may be added by referring to the amino acid content recommended in Annex A. The used L-amino acid monomer sources shall comply with GB14880 or Appendix B requirements. | ||||||
| Optional components | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Choline(mg) | 1.7 | 12 | 7.1 | 50.2 | ||
| Inositol(mg) | 1 | 9.5 | 4.2 | 39.7 | ||
| Taurine(mg) | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine(mg) | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid (% total fatty acid) | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) If docosahexaenoic acid (22:6 n-3) is supplemented to the infant formula, at least the same amount of Arachidonic acid (20:4 n-6) should be supplemented. Eicosapentaenoic acid (20:5 n-3) may exist in long chain unsaturated fatty acids, of which the total content should not exceed that of docosahexaenoic acid. 3) Total fatty acid refers to the sum of C4 ~ C24 fatty acids. |
|
| Arachidonic acid (% total fatty acid) | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Items | Limits | ||||
| Moisture /(%) | ≤5.0 | |||||
| Ash /(%) | ≤4.0 | |||||
| Impurities /(mg/kg) | ≤12 | |||||
| Limits of contaminants | Items | Limits | ||||
| Lead (Pb) /(mg/kg) | ≤0.15 | |||||
| Nitrate (based on NaNO3) /(mg/kg) | ≤100 | |||||
| Nitrite (based on NaNO2) /(mg/kg) | ≤2.0 | |||||
| Tin (Sn) /(mg/kg) | ≤50 | Only limited to food packaged in tin plate containers. | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | ≤0.5 | ||||
| Limits of microorganisms | Item | Limits (if not otherwise specified, in CFU/g(ml)) | n: total sample number in 1 batch c: maximum tolerance number of sample, of which testing result between m and M. m: microbiological acceptance level M: microbiological maximum safety tolerance |
|||
| n | c | m | M | |||
| Aerobic plate count | 5 | 2 | 1000 | 10000 | Not applicable to products supplemented with active bacteria (aerobic and anaerobic probiotics) [the number of active bacteria of active probiotics in products should be ≥106 CFU/g(ml)]. | |
| Coliform bacteria | 5 | 2 | 10 | 100 | ||
| Staphylococcus aureus | 5 | 2 | 10 | 100 | ||
| Enterobacter sakazakii | 3 | 0 | 0/100g | – | Only applicable to formulas for infants of 0-6 months old. | |
| Salmonella | 5 | 0 | 0/25g | – | ||
| Urease activity | Items | Index | Products containing soy component should be consistent with this provision. | |||
| Qualitative determination of urease activity | Negative | |||||
| Annex A- Recommendation on essential and semi-essential Amino Acids Used in Infant Formula |
The essential and semi-essential amino acids in infant formula are recommended not less than the recommended level in table below. | By reference to the representative data that have been published related to essential and semi-essential amino acids and nitrogen content and/or protein content in human milk in China and considering a certain range of variation, the lower limit of essential and semi-essential amino acids in infant formula can be calculated (mg/g N). | ||||
| In the calculations, the concentration of tyrosine and phenylalanine can be added; if the proportion of methionine to cysteine is less than 2:1, the content can be added also. | ||||||
| Amino acid | Index | |||||
| mg/g N | mg/100kcal | |||||
| Cystine | 80 | 24.1 | ||||
| Histidine | 120 | 36.1 | ||||
| Isoleucine | 300 | 90.2 | ||||
| Leucine | 540 | 162.4 | ||||
| Lysine | 350 | 105.3 | ||||
| Methionine | 65 | 19.6 | ||||
| Phenylalanine | 180 | 54.1 | ||||
| Threonine | 250 | 75.2 | ||||
| Tryptophan | 110 | 33.1 | ||||
| Tyrosine | 200 | 60.2 | ||||
| Valine | 310 | 93.2 | ||||
Link to download the standard?
English version | Chinese version
Who can help for further guidance on compliance?
Accestra
Just like the Australia New Zealand Food Standards Code in Australia, the national food safety standards (GB standards) are counterparts in China. Usually, each category of foods has one corresponding GB product standard, which stipulates the definitions, sensory and physicochemical indexes, limits of contaminants, fungaltoxin, pesticides and microbial indicators of the food. The Chinese authority will conduct inspection and quality control based on related GB standards when the products arrive at China ports. If the tested product cannot meet the requirements of GB standards, it will be returned or destroyed. So making sure our product comply with corresponding GB standard is very important. GB 10765-2010 National Food Safety Standard Infant Formula is the GB product standard for infant formulas.
Abstract of standard
| Application Scope | This standard applies to infant formulas. | |||||
| Terms and definitions | Infants | Refer to persons of 0 ~ 12 months old. | ||||
| Milk-based liquid Infant formula | Refers to liquid products made only through physical methods, of which the main material is milk and milk protein products, supplemented with a proper amount of vitamin, minerals and other supplementary materials, which are applicable to normal infants, where the energy and nutrition can satisfy the requirements of growth and development of normal infants of 0~6 months old. | |||||
| Requirements for Raw Materials | Ingredients and food additives adopted should not contain gluten. | |||||
| Hydrogenated oil and fat should NOT be used. | ||||||
| Raw and supplementary materials treated by irradiation should NOT be used. | ||||||
| For milk-based infant formula, the preferred carbohydrate should be lactose, or lactose and the polymer of glucose. Only after pre-gelatinization, the starch can be added into the infant formula. Fructose can NOT be used. | ||||||
| Sensory requirements | Items | Requirements | ||||
| Color | Should conform to the features of related products. | |||||
| Taste and odor | Should conform to the features of related products. | |||||
| Structural state | Should conform to the features of related products. No visible foreign substances should be included in the product. | |||||
| Dissolvability | Should conform to the features of related products. | |||||
| Essential components | All essential components in the product is essential for the growth and development of infants. | |||||
| The energy in the ready-to-eat infant formulas per 100ml should be within the range of 250 kJ (60 kcal)-295 kJ (70 kcal). The calculation of energy should be the value of the product of the content of protein, fat and carbohydrate per 100 mL product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber should be based on 50% of carbohydrate energy coefficient) respectively, and the sum (kJ/100mL) is divided by 4.184 to kcal/100 mL. | ||||||
| The protein, fat, carbohydrate content of infant formula per 100 kJ (100 kcal) should be consistent with the provisions set out in table below. | ||||||
| Nutrient | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Protein/(g) | 0.45 | 0.70 | 1.88 | 2.93 | For milk based infant formulas, the content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | 1.05 | 1.40 | 4.39 | 5.86 | Among the finished products, the amount of lauric acid and myristic acid (tetradecanoic acid) should account for no more than 20% of the total fatty acid; the maximum content of trans fatty acid should not exceed 3% of the total fatty acid; the erucic acid content should not exceed 1% of the total fatty acid. The total fatty acid refers to the sum of C4~C24 fatty acid. | |
| In which: linoleic acid(g) | 0.07 | 0.33 | 0.29 | 1.38 | ||
| α- linolenic acid (mg) | 12 | N.S. | 50 | N.S. | N.S.: No specification | |
| Linoleic acid/α- linolenic acid ratio | 5:1 | 15:1 | 5:1 | 15:1 | ||
| Total carbohydrate /(g) | ≤5.0 | The content of lactose in total carbohydrate should be over or equal to 90%. For calculation of the proportion of lactose among the total carbohydrates, the added oligosaccharides and polysaccharides should not be included. | ||||
| Carbohydrate content A1 should be calculated according to formula(1): A1= 100 − (A2+ A3+ A4+ A5+ A6) ………………………………(1) Where: A1——carbohydrate content, g/100g; A2——protein content, g/100g; A3——fat content, g/100g; A4——moisture content, g/100g; A5——ash content, g/100g; A6——dietary fiber content, g/100g. |
||||||
| Vitamins | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Vitamin A /(μg RE) | 14 | 43 | 59 | 180 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | 0.25 | 0.60 | 1.05 | 2.51 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | 0.12 | 1.20 | 0.50 | 5.02 | 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3). | |
| Vitamin K1/(μg) | 1.0 | 6.5 | 4.2 | 27.2 | ||
| Vitamin B1/(μg) | 14 | 72 | 59 | 301 | ||
| Vitamin B2/(μg) | 19 | 119 | 80 | 498 | ||
| Vitamin B6/(μg) | 8.5 | 45.0 | 35.6 | 188.3 | ||
| Vitamin B12/(μg) | 0.025 | 0.360 | 0.105 | 1.506 | ||
| Niacin (niacinamide)/(μg) | 70 | 360 | 293 | 1506 | Niacin: excludes precursor form. | |
| Folic acid/(μg) | 2.5 | 12.0 | 10.5 | 50.2 | ||
| Pantothenic acid/(μg) | 96 | 478 | 402 | 2000 | ||
| Vitamin C /(mg) | 2.5 | 17.0 | 10.5 | 71.1 | ||
| Biotin/(μg) | 0.4 | 2.4 | 1.5 | 10.0 | ||
| Minerals | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Sodium/(mg) | 5 | 14 | 21 | 59 | ||
| Potassium/(mg) | 14 | 43 | 59 | 180 | ||
| Copper/(μg) | 8.5 | 29 | 35.6 | 121.3 | ||
| Magnesium/(mg) | 1.2 | 3.6 | 5 | 15.1 | ||
| Iron/(mg) | 0.1 | 0.36 | 0.42 | 1.51 | ||
| Zinc/(mg) | 0.12 | 0.36 | 0.5 | 1.51 | ||
| Manganese/(mg) | 1.2 | 24 | 5 | 100.4 | ||
| Calcium/(mg) | 12 | 35 | 50 | 146 | ||
| Phosphorus/(mg) | 6 | 24 | 25 | 100 | ||
| Calcium/phosphorus ratio | 1:1 | 2:1 | 1:1 | 2:1 | ||
| Iodine/(μg) | 2.5 | 14 | 10.5 | 58.6 | ||
| Chloride/(mg) | 12 | 38 | 50 | 159 | ||
| Selenium/(μg) | 0.48 | 1.9 | 2.01 | 7.95 | ||
| Optional components | In addition to the essential components in the table above, one or more nutrients listed in the table below can be selected to add or claimed on label, whereas the content of such nutrients should meet the specification of Table below. | |||||
| To improve the protein quality of infant formula or enhance its nutritional value, the monomer L-amino acid may be added by referring to the amino acid content recommended in Annex A. The used L-amino acid monomer sources shall comply with GB14880 or Appendix B requirements. | ||||||
| Optional components | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Choline(mg) | 1.7 | 12 | 7.1 | 50.2 | ||
| Inositol(mg) | 1 | 9.5 | 4.2 | 39.7 | ||
| Taurine(mg) | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine(mg) | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid (% total fatty acid) | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) If docosahexaenoic acid (22:6 n-3) is supplemented to the infant formula, at least the same amount of Arachidonic acid (20:4 n-6) should be supplemented. Eicosapentaenoic acid (20:5 n-3) may exist in long chain unsaturated fatty acids, of which the total content should not exceed that of docosahexaenoic acid. 3) Total fatty acid refers to the sum of C4 ~ C24 fatty acids. |
|
| Arachidonic acid (% total fatty acid) | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Items | Limits | ||||
| Ash (calculated by dry substance) /(%) | ≤4.2 | |||||
| Impurities /(mg/kg) | ≤2 | |||||
| Limits of contaminants | Items | Limits | ||||
| Lead (Pb) /(mg/kg) | ≤0.02 | as ready-to-usestate | ||||
| Nitrate (based on NaNO3) /(mg/kg) | ≤100 | calculated based on powder product | ||||
| Nitrite (based on NaNO2) /(mg/kg) | ≤2.0 | calculated based on powder product | ||||
| Tin (Sn) /(mg/kg) | ≤50 | Only limited to food packaged in tin plate containers. | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | ≤0.5 | calculated based on powder product | |||
| Limits of microorganisms | The liquid infant formulas should meet the requirement of commercial sterilization | |||||
| Urease activity | Items | Index | 1) Products containing soy component should be consistent with this provision; 2) The sampling quantity of liquid infant formulas should be converted according to the content of dry substances. |
|||
| Qualitative determination of urease activity | Negative | |||||
| Annex A- Recommendation on essential and semi-essential Amino Acids Used in Infant Formula |
The essential and semi-essential amino acids in infant formula are recommended not less than the recommended level in table below. | By reference to the representative data that have been published related to essential and semi-essential amino acids and nitrogen content and/or protein content in human milk in China and considering a certain range of variation, the lower limit of essential and semi-essential amino acids in infant formula can be calculated (mg/g N). | ||||
| In the calculations, the concentration of tyrosine and phenylalanine can be added; if the proportion of methionine to cysteine is less than 2:1, the content can be added also. | ||||||
| Amino acid | Index | |||||
| mg/g N | mg/100kcal | |||||
| Cystine | 80 | 24.1 | ||||
| Histidine | 120 | 36.1 | ||||
| Isoleucine | 300 | 90.2 | ||||
| Leucine | 540 | 162.4 | ||||
| Lysine | 350 | 105.3 | ||||
| Methionine | 65 | 19.6 | ||||
| Phenylalanine | 180 | 54.1 | ||||
| Threonine | 250 | 75.2 | ||||
| Tryptophan | 110 | 33.1 | ||||
| Tyrosine | 200 | 60.2 | ||||
| Valine | 310 | 93.2 | ||||
Link to download the standard?
English version | Chinese version
Who can help for further guidance on compliance?
Accestra
Just like the Australia New Zealand Food Standards Code in Australia, the national food safety standards (GB standards) are counterparts in China. Usually, each category of foods has one corresponding GB product standard, which stipulates the definitions, sensory and physicochemical indexes, limits of contaminants, fungaltoxin, pesticides and microbial indicators of the food. The Chinese authority will conduct inspection and quality control based on related GB standards when the products arrive at China ports. If the tested product cannot meet the requirements of GB standards, it will be returned or destroyed. So making sure our product comply with corresponding GB standard is very important. GB 10765-2010 National Food Safety Standard Older infants and young children formula is the GB product standard for older infants and young children formula.
Abstract of standard
| Application Scope | This standard applies to infant formulas. | |||||
| Terms and definitions | Older infants | Refer to persons of 6~ 12 months old. | ||||
| Young children | Refer to young children of 12 – 36 months old. | |||||
| Milk-based powder older infants and young children formula | Refer to powder products made only through physical methods, of which the main material is milk and its product, supplemented with a proper amount of vitamins, minerals and other supplementary materials, which are applicable to older infants and young children, where the nutrition can satisfy partial requirements normal older infants and young children. | |||||
| Requirements for Raw Materials | Hydrogenated oil and fat should NOT be used. | |||||
| Raw and supplementary materials treated by irradiation should NOT be used. | ||||||
| Sensory requirements | Items | Requirements | ||||
| Color | Should conform to the features of related products. | |||||
| Taste and odor | Should conform to the features of related products. | |||||
| Structural state | Should conform to the features of related products, no visible foreign matter. | |||||
| Dissolvability | Should conform to the features of related products. | |||||
| Essential components | All essential components used in products should be necessary for growth and development of older infants and young children. | |||||
| The energy in the ready-to-eat older infants and young children formula per 100ml should be within the range of 250 kJ (60 kcal)-355 kJ (85 kcal). For calculation of energy, the content of protein, fat, carbohydrate per 100mL of product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber is calculated as 50% of carbohydrate energy coefficient) respectively, the obtained sum (kJ/100mL) divided by 4.184 to obtain the kcal/100mL value. | ||||||
| Protein and fat contained per 100kJ (100kcal) ready-to-eat older infants and young children formula should meet the specification in table below. The content of trans fatty acid should not be more than 3% total fatty acid. | ||||||
| Nutrient | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Protein/(g) | 0.7 | 1.2 | 2.9 | 5.0 | the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | 0.7 | 1.4 | 2.9 | 5.9 | ||
| In which: linoleic acid /(g) | 0.07 | N.S. | 0.29 | N.S. | N.S.: No specification | |
| Vitamins | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Vitamin A /(μg RE) | 18 | 54 | 75 | 225 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | 0.25 | 0.75 | 1.05 | 3.14 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | 0.15 | N.S. | 0.63 | N.S. | 1) 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3); 2) N.S.: No specification |
|
| Vitamin K1/(μg) | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Vitamin B1/(μg) | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B2/(μg) | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B6/(μg) | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B12/(μg) | 0.04 | N.S. | 0.17 | N.S. | N.S.: No specification | |
| Niacin (niacinamide)/(μg) | 110 | N.S. | 460 | N.S. | 1) Niacin: excludes precursor form; 2) N.S.: No specification | |
| Folic acid/(μg) | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Pantothenic acid/(μg) | 70 | N.S. | 293 | N.S. | N.S.: No specification | |
| Vitamin C /(mg) | 1.8 | N.S. | 7.5 | N.S. | N.S.: No specification | |
| Biotin/(μg) | 0.4 | N.S. | 1.7 | N.S. | N.S.: No specification | |
| Minerals | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Sodium/(mg) | N.S. | 20 | N.S. | 84 | N.S.: No specification | |
| Potassium/(mg) | 18 | 69 | 75 | 289 | ||
| Copper/(μg) | 7 | 35 | 29 | 146 | ||
| Magnesium/(mg) | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Iron/(mg) | 0.25 | 0.5 | 1.05 | 2.09 | ||
| Zinc/(mg) | 0.1 | 0.3 | 0.4 | 1.3 | ||
| Calcium/(mg) | 17 | N.S. | 71 | N.S. | N.S.: No specification | |
| Phosphorus/(mg) | 8.3 | N.S. | 34.7 | N.S. | N.S.: No specification | |
| Calcium/phosphorus ratio | 1.2:1 | 2:1 | 1.2:1 | 2:1 | ||
| Iodine/(μg) | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Chloride/(mg) | N.S. | 52 | N.S. | 218 | N.S.: No specification | |
| Optional components | Besides the essential components specified in table above, one or more optional components as shown in table below can be added to or claimed in the label of older infants and young children formula, whereas the content should meet the specification of table below. | |||||
| Optional components | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Selenium /(μg) | 0.48 | 1.90 | 2.01 | 7.95 | ||
| Choline /(mg) | 1.7 | 12.0 | 7.1 | 50.2 | ||
| Manganese /(μg) | 0.25 | 24.0 | 1.05 | 100.4 | ||
| Inositol /(mg) | 1.0 | 9.5 | 4.2 | 39.7 | ||
| Taurine /(mg) | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine /(mg) | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid /(% total fatty acid) | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) Total fatty acid refers to the sum of C4-C24 fatty acids. |
|
| Arachidonic acid /(% total fatty acid) | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Items | Limits | ||||
| Moisture /(%) | ≤5.0 | |||||
| Ash /(%) | ≤5.0 | |||||
| Impurities /(mg/kg) | ≤12 | NOT applicable to products supplemented with fruits and vegetables. | ||||
| Limits of contaminants | Items | Limits | ||||
| Lead (Pb) /(mg/kg) | ≤0.15 | |||||
| Nitrate (based on NaNO3) /(mg/kg) | ≤100 | NOT applicable to products supplemented with fruits and vegetables | ||||
| Nitrite (based on NaNO2) /(mg/kg) | ≤2.0 | |||||
| Tin (Sn) /(mg/kg) | ≤50 | Only limited to food packaged in tin plate containers. | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | ≤0.5 | ||||
| Limits of microorganisms | Item | Limits (if not otherwise specified, in CFU/g(ml)) | n: total sample number in 1 batch c: maximum tolerance number of sample, of which testing result between m and M. m: microbiological acceptance level M: microbiological maximum safety tolerance |
|||
| n | c | m | M | |||
| Aerobic plate count | 5 | 2 | 1000 | 10000 | Not applicable to products supplemented with active bacteria (aerobic and anaerobic probiotics) [the number of active bacteria of active probiotics in products should be ≥106 CFU/g(ml)]. | |
| Coliform bacteria | 5 | 2 | 10 | 100 | ||
| Salmonella | 5 | 0 | 0/25g | – | ||
| Urease activity | Items | Index | Products containing soy component should be consistent with this provision. | |||
| Qualitative determination of urease activity | Negative | |||||
Application ScopeThis standard applies to infant formulas.
Terms and definitionsInfantsRefer to persons of 0 ~ 12 months old.
Milk-based liquid Infant formulaRefers to liquid products made only through physical methods, of which the main material is milk and milk protein products, supplemented with a proper amount of vitamin, minerals and other supplementary materials, which are applicable to normal infants, where the energy and nutrition can satisfy the requirements of growth and development of normal infants of 0~6 months old.
Requirements for Raw MaterialsIngredients and food additives adopted should not contain gluten.
Hydrogenated oil and fat should NOT be used.
Raw and supplementary materials treated by irradiation should NOT be used.
For milk-based infant formula, the preferred carbohydrate should be lactose, or lactose and the polymer of glucose. Only after pre-gelatinization, the starch can be added into the infant formula. Fructose can NOT be used.
Sensory requirementsItemsRequirements
ColorShould conform to the features of related products.
Taste and odorShould conform to the features of related products.
Structural stateShould conform to the features of related products. No visible foreign substances should be included in the product.
DissolvabilityShould conform to the features of related products.
Essential componentsAll essential components in the product is essential for the growth and development of infants.
The energy in the ready-to-eat infant formulas per 100ml should be within the range of 250 kJ (60 kcal)-295 kJ (70 kcal). The calculation of energy should be the value of the product of the content of protein, fat and carbohydrate per 100 mL product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber should be based on 50% of carbohydrate energy coefficient) respectively, and the sum (kJ/100mL) is divided by 4.184 to kcal/100 mL.
The protein, fat, carbohydrate content of infant formula per 100 kJ (100 kcal) should be consistent with the provisions set out in table below.
NutrientLimits
Per 100 kJPer 100 kcal
MinimumMaximumMinimumMaximum
Protein/(g)0.450.701.882.93For milk based infant formulas, the content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25Fat/(g)1.051.404.395.86Among the finished products, the amount of lauric acid and myristic acid (tetradecanoic acid) should account for no more than 20% of the total fatty acid; the maximum content of trans fatty acid should not exceed 3% of the total fatty acid; the erucic acid content should not exceed 1% of the total fatty acid. The total fatty acid refers to the sum of C4~C24 fatty acid.In which: linoleic acid(g)0.070.330.291.38
α- linolenic acid (mg)12N.S.50N.S.N.S.: No specificationLinoleic acid/α- linolenic acid ratio5:115:15:115:1
Total carbohydrate /(g)≤5.0The content of lactose in total carbohydrate should be over or equal to 90%. For calculation of the proportion of lactose among the total carbohydrates, the added oligosaccharides and polysaccharides should not be included.Carbohydrate content A1 should be calculated according to formula(1):
A1= 100 − (A2+ A3+ A4+ A5+ A6) ………………………………(1)
Where:
A1——carbohydrate content, g/100g;
A2——protein content, g/100g;
A3——fat content, g/100g;
A4——moisture content, g/100g;
A5——ash content, g/100g;
A6——dietary fiber content, g/100g.VitaminsLimits
Per 100 kJPer 100 kcal
MinimumMaximumMinimumMaximum
Vitamin A /(μg RE)144359180RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A.Vitamin D /(μg)0.250.601.052.51Calciferol, 1μg Vitamin D = 40 IU Vitamin DVitamin E /(mg α-TE)0.121.200.505.021 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3).Vitamin K1/(μg)1.06.54.227.2
Vitamin B1/(μg)147259301
Vitamin B2/(μg)1911980498
Vitamin B6/(μg)8.545.035.6188.3
Vitamin B12/(μg)0.0250.3600.1051.506
Niacin (niacinamide)/(μg)703602931506Niacin: excludes precursor form.Folic acid/(μg)2.512.010.550.2
Pantothenic acid/(μg)964784022000
Vitamin C /(mg)2.517.010.571.1
Biotin/(μg)0.42.41.510.0
MineralsLimits
Per 100 kJPer 100 kcal
MinimumMaximumMinimumMaximum
Sodium/(mg)5142159
Potassium/(mg)144359180
Copper/(μg)8.52935.6121.3
Magnesium/(mg)1.23.6515.1
Iron/(mg)0.10.360.421.51
Zinc/(mg)0.120.360.51.51
Manganese/(mg)1.2245100.4
Calcium/(mg)123550146
Phosphorus/(mg)62425100
Calcium/phosphorus ratio1:12:11:12:1
Iodine/(μg)2.51410.558.6
Chloride/(mg)123850159
Selenium/(μg)0.481.92.017.95
Optional componentsIn addition to the essential components in table above, if one or more nutrients listed in Table below can be selected to add or claimed on label, whereas the content of such nutrients should meet the specification of Table below.
To improve the protein quality of infant formula or enhance its nutritional value, the monomer L-amino acid may be added by referring to the amino acid content recommended in Annex A. The used L-amino acid monomer sources shall comply with GB14880 or Appendix B requirements.
Optional componentsLimits
Per 100 kJPer 100 kcal
MinimumMaximumMinimumMaximum
Choline(mg)1.7127.150.2
Inositol(mg)19.54.239.7
Taurine(mg)N.S.3N.S.13N.S.: No specificationL-Carnitine(mg)0.3N.S.1.3N.S.N.S.: No specificationDocosahexaenoic acid (% total fatty acid)N.S.0.5N.S.0.51) N.S.: No specification;
2) If docosahexaenoic acid (22:6 n-3) is supplemented to the infant formula, at least the same amount of Arachidonic acid (20:4 n-6) should be supplemented. Eicosapentaenoic acid (20:5 n-3) may exist in long chain unsaturated fatty acids, of which the total content should not exceed that of docosahexaenoic acid.
3) Total fatty acid refers to the sum of C4 ~ C24 fatty acids.Arachidonic acid (% total fatty acid)N.S.1N.S.1Other indexesItemsLimits
Ash (calculated by dry substance) /(%)≤4.2
Impurities /(mg/kg)≤2
Limits of contaminantsItemsLimits
Lead (Pb) /(mg/kg)≤0.02as ready-to-usestateNitrate (based on NaNO3) /(mg/kg)≤100calculated based on powder productNitrite (based on NaNO2) /(mg/kg)≤2.0calculated based on powder productTin (Sn) /(mg/kg)≤50Only limited to food packaged in tin plate containers.Limits of mycotoxinsAflatoxin M1/(μg/kg)≤0.5calculated based on powder productLimits of microorganismsThe liquid infant formulas should meet the requirement of commercial sterilization
Urease activityItemsIndex1) Products containing soy component should be consistent with this provision;
2) The sampling quantity of liquid infant formulas should be converted according to the content of dry substances.Qualitative determination of urease activityNegativeAnnex A-
Recommendation on essential and semi-essential Amino Acids Used in Infant FormulaThe essential and semi-essential amino acids in infant formula are recommended not less than the recommended level in table below.By reference to the representative data that have been published related to essential and semi-essential amino acids and nitrogen content and/or protein content in human milk in China and considering a certain range of variation, the lower limit of essential and semi-essential amino acids in infant formula can be calculated (mg/g N).In the calculations, the concentration of tyrosine and phenylalanine can be added; if the proportion of methionine to cysteine is less than 2:1, the content can be added also.Amino acidIndexmg/g Nmg/100kcalCystine8024.1Histidine12036.1Isoleucine30090.2Leucine540162.4Lysine350105.3Methionine6519.6Phenylalanine18054.1Threonine25075.2Tryptophan11033.1Tyrosine20060.2Valine31093.2
Link to download the standard?
English version | Chinese version
Who can help for further guidance on compliance?
Accestra
Just like the Australia New Zealand Food Standards Code in Australia, the national food safety standards (GB standards) are counterparts in China. Usually, each category of foods has one corresponding GB product standard, which stipulates the definitions, sensory and physicochemical indexes, limits of contaminants, fungaltoxin, pesticides and microbial indicators of the food. The Chinese authority will conduct inspection and quality control based on related GB standards when the products arrive at China ports. If the tested product cannot meet the requirements of GB standards, it will be returned or destroyed. So making sure our product comply with corresponding GB standard is very important. GB 10765-2010 National Food Safety Standard Older infants and young children formula is the GB product standard for older infants and young children formula.
Abstract of standard
| Application Scope | This standard applies to infant formulas. | |||||
| Terms and definitions | Older infants | Refer to persons of 6~ 12 months old. | ||||
| Young children | Refer to young children of 12 – 36 months old. | |||||
| Milk-based liquid older infants and young children formula | Refer to liquid products made only through physical methods, of which the main material is milk and its product, supplemented with a proper amount of vitamins, minerals and other supplementary materials, which are applicable to older infants and young children, where the nutrition can satisfy partial requirements normal older infants and young children. | |||||
| Requirements for Raw Materials | Hydrogenated oil and fat should NOT be used. | |||||
| Raw and supplementary materials treated by irradiation should NOT be used. | ||||||
| Sensory requirements | Items | Requirements | ||||
| Color | Should conform to the features of related products. | |||||
| Taste and odor | Should conform to the features of related products. | |||||
| Structural state | Should conform to the features of related products, no visible foreign matter. | |||||
| Dissolvability | Should conform to the features of related products. | |||||
| Essential components | All essential components used in products should be necessary for growth and development of older infants and young children. | |||||
| The energy in the ready-to-eat older infants and young children formula per 100ml should be within the range of 250 kJ (60 kcal)-355 kJ (85 kcal). For calculation of energy, the content of protein, fat, carbohydrate per 100mL of product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber is calculated as 50% of carbohydrate energy coefficient) respectively, the obtained sum (kJ/100mL) divided by 4.184 to obtain the kcal/100mL value. | ||||||
| Protein and fat contained per 100kJ (100kcal) ready-to-eat older infants and young children formula should meet the specification in table below. The content of trans fatty acid should not be more than 3% total fatty acid. | ||||||
| Nutrient | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Protein/(g) | 0.7 | 1.2 | 2.9 | 5.0 | the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | 0.7 | 1.4 | 2.9 | 5.9 | ||
| In which: linoleic acid /(g) | 0.07 | N.S. | 0.29 | N.S. | N.S.: No specification | |
| Vitamins | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Vitamin A /(μg RE) | 18 | 54 | 75 | 225 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | 0.25 | 0.75 | 1.05 | 3.14 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | 0.15 | N.S. | 0.63 | N.S. | 1) 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3); 2) N.S.: No specification |
|
| Vitamin K1/(μg) | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Vitamin B1/(μg) | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B2/(μg) | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B6/(μg) | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B12/(μg) | 0.04 | N.S. | 0.17 | N.S. | N.S.: No specification | |
| Niacin (niacinamide)/(μg) | 110 | N.S. | 460 | N.S. | 1) Niacin: excludes precursor form; 2) N.S.: No specification | |
| Folic acid/(μg) | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Pantothenic acid/(μg) | 70 | N.S. | 293 | N.S. | N.S.: No specification | |
| Vitamin C /(mg) | 1.8 | N.S. | 7.5 | N.S. | N.S.: No specification | |
| Biotin/(μg) | 0.4 | N.S. | 1.7 | N.S. | N.S.: No specification | |
| Minerals | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Sodium/(mg) | N.S. | 20 | N.S. | 84 | N.S.: No specification | |
| Potassium/(mg) | 18 | 69 | 75 | 289 | ||
| Copper/(μg) | 7 | 35 | 29 | 146 | ||
| Magnesium/(mg) | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Iron/(mg) | 0.25 | 0.5 | 1.05 | 2.09 | ||
| Zinc/(mg) | 0.1 | 0.3 | 0.4 | 1.3 | ||
| Calcium/(mg) | 17 | N.S. | 71 | N.S. | N.S.: No specification | |
| Phosphorus/(mg) | 8.3 | N.S. | 34.7 | N.S. | N.S.: No specification | |
| Calcium/phosphorus ratio | 1.2:1 | 2:1 | 1.2:1 | 2:1 | ||
| Iodine/(μg) | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Chloride/(mg) | N.S. | 52 | N.S. | 218 | N.S.: No specification | |
| Optional components | Besides the essential components specified in table above, one or more optional components as shown in table below can be added to or claimed in the label of older infants and young children formula, whereas the content should meet the specification of table below. | |||||
| Optional components | Limits | |||||
| Per 100 kJ | Per 100 kcal | |||||
| Minimum | Maximum | Minimum | Maximum | |||
| Selenium /(μg) | 0.48 | 1.90 | 2.01 | 7.95 | ||
| Choline /(mg) | 1.7 | 12.0 | 7.1 | 50.2 | ||
| Manganese /(μg) | 0.25 | 24.0 | 1.05 | 100.4 | ||
| Inositol /(mg) | 1.0 | 9.5 | 4.2 | 39.7 | ||
| Taurine /(mg) | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine /(mg) | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid /(% total fatty acid) | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) Total fatty acid refers to the sum of C4-C24 fatty acids. |
|
| Arachidonic acid /(% total fatty acid) | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Items | Limits | ||||
| Ash (calculated by dry substance) /(%) | ≤5.3 | |||||
| Impurities /(mg/kg) | ≤2 | NOT applicable to products supplemented with fruits and vegetables. | ||||
| Limits of contaminants | Items | Limits | ||||
| Lead (Pb) /(mg/kg) | ≤0.02 | as ready-to-usestate | ||||
| Nitrate (based on NaNO3) /(mg/kg) | ≤100 | 1) NOT applicable to products supplemented with fruits and vegetables; 2) calculated based on powder product. | ||||
| Nitrite (based on NaNO2) /(mg/kg) | ≤2.0 | calculated based on powder product | ||||
| Tin (Sn) /(mg/kg) | ≤50 | Only limited to food packaged in tin plate containers. | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | ≤0.5 | calculated based on powder product | |||
| Limits of microorganisms | The liquid product should meet the requirement of commercial sterilization | |||||
| Urease activity | Items | Index | 1) Products containing soy component should be consistent with this provision; 2) The sampling quantity of liquid infant formulas should be converted according to the content of dry substances. |
|||
| Qualitative determination of urease activity | Negative | |||||
Link to download the standard?
English version | Chinese version
Who can help for further guidance on compliance?
Accestra
Introduction to Chinese Labelling
According to “Food Safety Law of China”, pre-packaged food should be labeled on the package. Imported prepackaged food and food additive shall have Chinese labels.
In China, there is a general standard for the labelling of prepackaged normal foods which is called GB 7718. This standard applies to the labelling of prepackaged normal foods available directly and indirectly to consumers.
In addition, there is another labelling standard of prepackaged foods for special dietary purpose which is called GB 13432. Foods for special dietary purpose include:
─ Infant formula
─ Older infants and young children formula
─ Infant formula for special medical purpose
─ Cereal-based complementary foods for infants and young children
─ Canned complementary foods for infants and young children
─ Food for special medical purpose (excluding Infant formula for special medical purpose)
─ Others
Besides the above labeling standards, to be a qualified Chinese label, it should also comply with the corresponding product standard if there is any requirement for the labeling in the product standard.
In China, there are two forms of labeling:
─ Paste a Chinese label on the original label (Pic 1). Usually, the pasting work can be done before arriving at the port or in the specified warehouse in the bonded area.
Note: Infant formula should have the Chinese label designed and printed on the packaging directly before importation, the label of infant formula cannot be pasted on the original label.
─ Design a Chinese label and print on the packaging directly (Pic 2). You can also use foreign languages simultaneously in corresponding to Chinese (excluding trademark, name and address of overseas manufacturers, name and address of overseas distributors, website). All foreign characters shall not be larger than the corresponding Chinese characters (except for trademark).

You cannot make claim of any contents related to the function of disease prevention and treatment in food labels.
According to GB 7718, the mandatory items for labels of imported normal foods are as follows:
Product name, ingredient list, net content, importer information, country of origin, manufacturing date, shelf life, storage condition, nutrition information.
Other information, such as allergen, consumption method are recommended items, they can be labelled optionally.
In the Chinese label, the font size of all Chinese characters should be no less than 1.8mm. For net content, the font size should meet the requirements below:
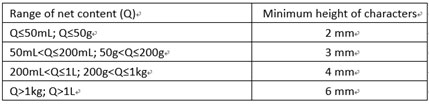
According to GB 13432, labelling of food for special dietary purpose should not only meet the basic requirements in GB 7718, but also should comply with relevant provisions for labels in the corresponding product standards. In addition, consumption methods and suitable groups should also be indicated on the label.
According to GB 28050 “General rules for nutrition labelling of prepackaged normal foods”, while meeting certain requirements, you can make claim to the content of energy or nutrients on the label, for example:
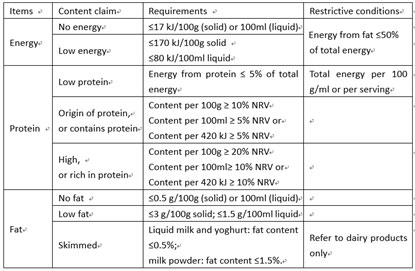
According to GB 28050, while meeting the requirements of content claims, you can make claim to the function of energy or nutrients on the label. However, you can only use the claims listed in Appendix D of GB 28050 and the words cannot be deleted, added or combined.
According to the requirement from Customs, the label of imported dairy products should also indicate CNCA registration number of overseas manufacturer.
How to design a Chinese label?
Step 1
Identify the category:
Infant formula belongs to food for special dietary purpose, and therefore, the Chinese label must be printed directly on the packaging before exportation.
Step 2
Find the general GB standard:
The general GB standard for the labelling of infant formula is GB 13432, which stipulates the general rules for infant formula labels.
According to GB 13432, the Chinese label should at least contain: Product name, ingredient list, net content, importer information, country of origin, manufacturing date, shelf life, storage condition, nutrition information, consumption methods and suitable groups.
Please notice that infant formula for 0-6 month-old infants are not allowed to make any claim to the content or function of the essential nutrients.
Step 3
Find the corresponding product standard:
The product standard for infant formula is GB 10765. In GB 10765, there are some additional requirements for the labelling of infant formula:
- In nutrition information, the content of nutrients as per 100kJ should also be indicated.
- Category of products, properties of infant formula (for example, milk-based/bean-based and states of products) and age range shall be labelled. Infant formula for infants older than 6 months-old shall indicate the notice of “6个月龄以上婴儿食用本产品时,应配合添加辅助食品” (For infants older than 6 months, supplementary foods shall be used).
- Labels of the infant formula shall include the notice of “对于0-6月的婴儿最理想的食品是母乳,在母乳不足或无母乳时可食用本产品” (The best treatment for infants of 0-6 month-old is breast milk, this product can be used to fulfil infants’ needs when mothers are unable to supply sufficient volumes of breast milk).
- Images of infants and women are not allowed on labels, expressions such as humanised milk and maternalised milk shall not be used.
- The directions for use, proper preparation and illustration of the product shall be clearly labelled. Warnings on potential health harms caused by improper mixing or use shall be indicated in the directions for use.
Step 4
Find whether there are any other regulations regarding infant formula labelling:
There is a regulation called “Infant formula registration-Technical guidelines for the labelling (trial)” (“Guidelines” for short) which stipulates the detailed requirements for infant formula labels. According to the guidelines, the labels of infant formula should not only include the above contents, but also comply with the requirements below:
- The registration number of infant formula should be indicated.
- The CNCA registration number of overseas manufacturers should be indicated.
- The principal display panel of label shall be indicated with product name, net content, registration number. The registered trademark can be indicated on the principal display panel as well, but if the trademark is not one part of the product name, it should be labelled on the corner of the label.
- Content claim and functional claim should be indicated in the form of text on non-principal display panel.
Step 5
So the label contents of imported infant formula should include:
Product name, ingredient list, net content, importer information, country of origin, manufacturing date, shelf life, storage condition, nutrition information, consumption methods/directions for use, suitable groups, overseas manufacturer CNCA registration number, infant formula registration number, product category, properties of infant formula (milk-based, states of products), warnings and notice.
Samples

Top Tips
- The label should be printed on the packaging before exportation.
- Please make sure that all mandatory items are included on the label.
- Please make sure all Chinese characters are larger than 1.8 mm (except the net content, please choose the corresponding font size according to the net content).
- When making claims of any nutrients, please make sure they can meet corresponding requirements. For infant formula, such claims can only be indicated on the non-principal display panel, and infant formula for 0-6 month-old infants are not allowed to make any claim to the content or function of the essential nutrients.
- “Nature”, “no add preservatives”, “no add additives” cannot be indicated on the label.
How to design a Chinese label?
Step 1
Identify the category:
Older infants formula belongs to food for special dietary purpose, and particularly, the Chinese label should be printed directly on the packaging before exportation.
Step 2
Find the general GB standard:
The general GB standard for the labelling is GB 13432, which stipulates the general rules for older infants formula labels.
According to GB 13432, the Chinese label should at least contain: Product name, ingredient list, net content, importer information, country of origin, manufacturing date, shelf life, storage condition, nutrition information, consumption methods and suitable groups.
Step 3
Find the corresponding product standard:
The product standard for older infants formula is GB 10767. In GB 10767, there are some additional requirements for the labelling of older infants formula:
-
- In nutrition information, the content of nutrients as per 100kJ should also be indicated.
- Product category, product properties (for example, milk-based/bean-based and states of products) and age range shall be labeled. Older infants formula shall indicate the notice of “须配合添加辅助食品” (supplementary foods shall be used).
- The directions for use, proper preparation and illustration of the product shall be clearly labelled. Warnings on potential health harms caused by improper mix or use shall be indicated in the directions for use.
Step 4
Find whether there are any other regulations regarding older infants formula labelling:
There is a regulation called “Infant formula registration-Technical guidelines for the labelling (trial)” (“Guidelines” for short) which stipulates the detailed requirements for older infants formula labels. According to the guidelines, the labels of infant formula should not only include the above contents, but also comply with the requirements below:
-
-
- The registration number of older infants formula should be indicated.
- The CNCA registration number of overseas manufacturers should be indicated.
- The principal display panel of label shall be indicated with product name, net content, registration number. The registered trademark can be indicated on the principal display panel as well, but if the trademark is not one part of the product name, it should be labelled on the corner of the label.
- Content claim and functional claim should be indicated in the form of text on non-principal display panel.
-
Step 5
So the label contents of imported older infants formula should include:
Product name, ingredient list, net content, importer information, country of origin, manufacturing date, shelf life, storage condition, nutrition information, consumption methods/directions for use, suitable groups, overseas manufacturer CNCA registration number, infant formula registration number, product category, properties of infant formula (milk-based, states of products), warnings and notice.
Samples

Top Tips
-
-
- The label should be printed on the packaging before exportation.
- Please make sure that all mandatory items are included on the label.
- Please make sure all Chinese characters are larger than 1.8 mm (except the net content, please choose the corresponding font size according to the net content).
- When make claims of any nutrients, please make sure they can meet corresponding requirements. For older infants formula, such claims can only be indicated on the non-principal display panel.
- “Nature”, “no add preservatives”, “no add additives” cannot be indicated on the label.
-
How to design a Chinese label?
Step 1
Identify the category:
Young children formula belongs to food for special dietary purpose, and particularly, the Chinese label should be printed directly on the packaging before exportation.
Step 2
Find the general GB standard:
The general GB standard for the labelling is GB 13432, which stipulates the general rules for young children formula labels.
According to GB 13432, the Chinese label should at least contain: Product name, ingredient list, net content, importer information, country of origin, manufacturing date, shelf life, storage condition, nutrition information, consumption methods and suitable groups.
Step 3
Find the corresponding product standard:
The product standard for young children formula is GB 10767. In GB 10767, there are some additional requirements for the labelling of young children formula:
-
-
- In nutrition information, the content of nutrients as per 100kJ should also be indicated.
- Product category, product properties (for example, milk-based/bean-based and states of products) and age range shall be labeled.
- The directions for use, proper preparation and illustration of the product shall be clearly labelled. Warnings on potential health harms caused by improper mix or use shall be indicated in the directions for use.
-
Step 4
Find whether there are any other regulations regarding infant formula labelling:
There is a regulation called “Infant formula registration-Technical guidelines for the labelling (trial)” (“Guidelines” for short) which stipulates the detailed requirements for young children formula labels. According to the guidelines, the labels should not only include the above contents, but also comply with the requirements below:
-
-
-
- The registration number of older infants formula should be indicated..
- The CNCA registration number of overseas manufacturers should be indicated.
- The principal display panel of label shall be indicated with product name, net content, registration number. The registered trademark can be indicated on the principal display panel as well, but if the trademark is not one part of the product name, it should be labelled on the corner of the label.
- Content claim and functional claim should be indicated in the form of text on non-principal display panel.
-
-
Step 5
So the label contents of imported older infants formula should include:
Product name, ingredient list, net content, importer information, country of origin, manufacturing date, shelf life, storage condition, nutrition information, consumption methods/directions for use, suitable groups, overseas manufacturer CNCA registration number, young children formula registration number, product category, properties (milk-based, states of products), warnings and notice.
Samples
Top Tips
-
-
-
- The label should be printed on the packaging before exportation.
- Please make sure that all mandatory items are included on the label.
- Please make sure all Chinese characters are larger than 1.8 mm (except the net content, please choose the corresponding font size according to the net content).
- When make claims of any nutrients, please make sure they can meet corresponding requirements. For young children formula, such claims can only be indicated on the non-principal display panel.
- “Nature”, “no add preservatives”, “no add additives” cannot be indicated on the label.
-
-
Organic Certification
Australian Organic Certification currently does not have Mutual Recognition Agreement with China and if you would like to claim and export organic products, the product has to be certified to Chinese Organic Regulations. China Organic Certification are issued by an accredited certification body in China, while the requirements are similar to Australian Certified Organic Certification, the process and paper work requirements are different. To certify organic, the entire supply chain has to be Chinese certified which means the raw material supplies through to processing and manufacturing will have to be certified.
Organic products are products for human and animal consumption, where production, processing and marketing activities are in line with the China National Standards for Organic Products (GB/T 19630).
Organic certification: the conformity assessment activities where certification bodies, based on the provisions of management measures for certification of organic products and organic certification rules, prove that the production, processing and marketing activities of related products are in line with the China National Standards for Organic Products.
China has a positive list issued by CNCA which illustrates the scope of food categtories permitted for organic certification. Therefore, only products listed in the positive list and meet reqirements can apply for China orangic certification.
For dairy products, product categories in positive list permitted for organic certification are as follows
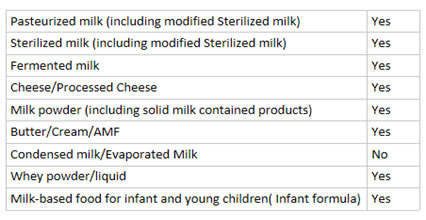
Procedures & Estimated timeline and costs
Procedures
- Applicant prepare dossier
- Certification body check dossier
- Certification body on-site inspection
- Certification body submit inspection report to CNCA
- CNCA issue certification number
- Certification body issue certificate to Applicant
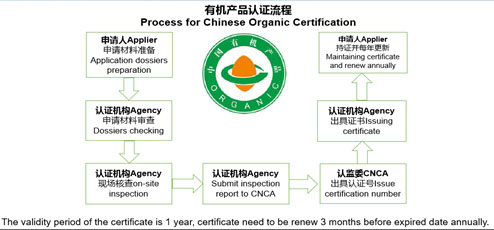
Timeline
If all application materials are completed accurately and all ingredients are certified organic, then the certification timeline is around 2-3 months. The on-site audits can be arranged at a convenient date with the auditor. The validity period of the certificate is one (1) year, so it needs to be renewed annually.
Please note the timeline will depend on various factors which could include: the status of certification for entire supply chain including raw materials & recipe, documentation from suppliers, sample record of inputs pest control, quality management, manufacturing processes and other conditions. Therefore a proper evaluation of the status of the applicant is required to assess a timetable for implementing China Organic Certification.
Costs
The cost of organic certification consists of 4 parts:
- Organic certification
- Testing fee
- Traveling and inspection cost of audit
- Consulting fee for certification authority
Please contact an accredited certification body for detailed break down on costings for China organic certification.
Link to responsible agencies and contacts
The competent authority of organic certification is CNCA. CNCA authorizes inspection and certification competency to its approved certification body (approx. 66 independent certification bodies).
Click this link: http://ffip.cnca.cn/ffip/publicquery/certSearch.jsp, you will be guided to the “China food and agricultural products certification information system”.
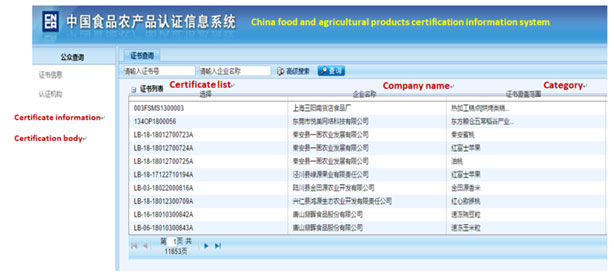
Click “Certification body”, then choose “Business scope” as the last one “Organic products” and click “Search”.
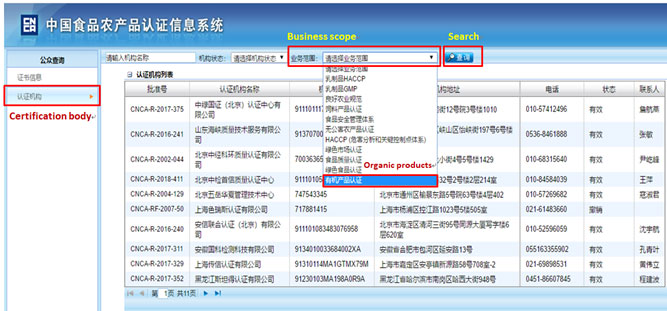
Choose one certification body from the list.
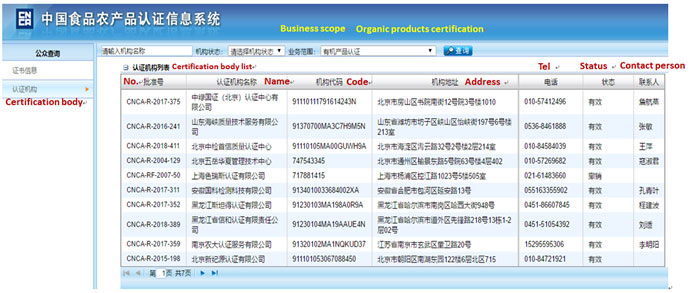
Australian based certified bodies
Australia’s largest internationally recognised certification body, Australian Certified Organic (ACO), and National Association for Sustainable Agriculture Australia Limited (NASSA), have both signed agreements with a Chinese certifier (NASSA media release | ACO media release) to provide Australian certified operators with a more accessable and convenient certified organic food certificate application.
Health Certificate
The Health certificate for dairy products exporting to China is a certificate issued by competent authority of exporting country and authorised by China Customs. In Australia, the issuing department is Department of Agriculture and Water Resources, DAWR. China Customs is responsible for inspection and accreditation of import and export food in China.
Health certificate should include following contents:
- Dairy raw materials were taken from healthy
- Dairy products have been prepared or processed and will not infect or carry any animal epidemic disease;
- The dairy manufacturing enterprise is under supervision of local government;
- The dairy products are safe and fit for human consumption.
Health certificate should be sealed and signed by local competent authority (Department of Agriculture and Water Resources).
According to “Announcement on standardizing the management of health certificate for imported dairy products”, imported dairy products without a health certificate will not be accepted from March 1, 2010.
Every exporting country that can export dairy products to China will have a different template of health certificate authorised by China Customs. While exporting your dairy products to China, you should make sure that the format of health certificate should be identical to the template on the China Customs website. (See Pic.1 for the health certificate template of Australia, the stamps and seals have been changed since 20 November 2016.)

Do you require health certificate for your product?
All dairy products exported to China will require a Health Certificate. So, to export dairy products to China, you need to obtain the health certificate from DAWR.
If so, who and how to obtain?
Who?
The exporter in Australia should obtain a health certificate from DAWR.
How?
- If the products met all requirements of Australia and China and are ready for export, you can make a Request for Permit (RFP) in the Export Documentation System (EXDOC).
- To make a RFP, you should have been registered as an exporter and EDI user in EXDOC. This can also be done by an agent on your behalf, if your detailed information is registered in EXDOC). Click here to find more information about EXDOC.
- While making a RFP, you also need to include details of dairy product codes in EXDOC.
- After the RFP is approved, you will be issued with a health certificate.
- Check the pricing per document fee for the health certificate.
Links to responsible agencies and contacts
Department of Agriculture and Water Resources
Website: http://www.agriculture.gov.au/export/controlled-goods/dairy
Telephone General inquiry: 1800 900 090 or + 61 3 8318 6700 (from outside Australia)
National office: +61 2 6272 3933
What is Certificate Of Origin (COO)?
Certificate of Origin (COO) is a document to certify the place of growth, production or manufacture of goods. Certificate of Australian Origin is a document to specify goods that have been grown, produced or manufactured in Australia (see Pic.1 for the sample).
COO is issued according to the requirements of the destination country, and can be classified as two types: Preferential and Non-Preferential Certificates. Preferential Certificates can only be issued when there is a Free Trade Agreement between the two countries, otherwise, Non-Preferential Certificates will be issued. The Customs of destination country will assess tariff treatment according to Preferential Certificates.
Since Australia and China has signed the China Australia Free Trade Agreement (ChAFTA), exporter of Australia may obtain Preferential Certificates.
Is it required for your product?
Although there are no regulation that explicitly stipulates COO is mandatory for dairy products in China, importers in China will always ask overseas exporters to provide COO for Customs clearance as common practice. The China Customs will use COO for trade statistics, sanctions and quota applications, antidumping administration, as well as the preferential tariff treatments.
In addition, since Australia and China has signed the ChAFTA, if you don’t provide COO, you cannot enjoy the agreement tariff under ChAFTA. Generally, the agreement tariff will be lower than general tariff rate and the ‘Most Favoured Nnation’ (MFN) rate.
As a result, COO is required and you need to provide it to the importers.
Please note that a COO only applies to a single shipment, it may cover one or more goods, but should not exceed 20 items (20 unique goods) and will remain valid for one year.
If so, who and how to obtain?
Who?
The exporter in Australia should obtain COO and provide it to the importer in China.
How?
Australian Chamber of Commerce and Industry (ACCI) and Australian Industry Group (AIG) are authorised to issue Certificates of Australian Origin for dairy products by the Australian Government, and there are several local accredited bodies who can issue COO on behalf of ACCI. The exporter can also obtain COO from those local accredited bodies.
There are some points that you should pay attention to:
- The Exporters Information Form Update must be completed and forwarded to the issuing body of COO, together with a list of signatories authorised to sign the certificates on behalf of your company.
- Evidence of origin must be supplied prior to stamping (such as copies of invoice, bill of lading, letter of credit, or statutory declaration).
- Exporters must provide a copy of the documents being stamped for Chamber records.
- All export documents must be signed by the exporter on the bottom left hand side under the exporter’s declaration before submission for authentication.
- Please note that Certificate of Australian Origin forms cannot be used for any other origin than Australian.
- Certificates of Origin must be typed.
Register COO today? Here are links to responsible agencies and contacts
| National | The Australian Chamber of Commerce and Industry (ACCI) Link: www.acci.asn.au/certificates-origin |
| National | The Australian Industry Group (AIG) Link: www.aigroup.com.au/business-services/trade/certificateoforigin/ |
| NSW | NSW Business Chamber Link: www.nswbusinesschamber.com.au |
| QLD | Chamber of Commerce & Industry Queensland Link: www.cciq.com.au |
| VIC | Victorian Chamber of Commerce Link: www.victorianchamber.com.au |
| SA | Business SA Link: www.business-sa.com |
| WA | Chamber of Commerce & Industry Western Australia Link: www.cciwa.com |
| NT | Chamber of Commerce Northern Territory Link: www.chambernt.com.au |
| TAS | Tasmanian Chamber of Commerce and Industry Link: www.tcci.com.au |
| ACT | Canberra Business Chamber Link: www.canberrabusiness.com |
| AACCI | Australian-Arab Chamber of Commerce and Industry Link: www.austarab.com.au |
Licence for quarantine of entry of animals and plants
According to “Administrative measures for the examination and approval of entry animal and plant quarantine”, in order to prevent the infection of zoonosis, parasitosis, pests and weeds of plants, plant and animal products should get the quarantine licenses. General Administration of Customs will publish the list of products that require quarantine licenses.
For dairy products, there are 3 categories that require quarantine licenses:
| No. | Categories | Relevant HS codes |
| 1 | Raw milk (Ordinary milk from healthy dairy stock without any ingredient change) |
0401200000 |
| 2 | Raw milk products (Dairy products using raw milk as the ingredient, without any heat-treatment or sterilization) |
0403100000, 0406100000, 0406200000, 0406300000, 0406400000, 0406900000 |
| 3 | Pasteurized milk, modified milk with pasteurization process (Only for liquid dairy products using raw milk as the ingredient, with pasteurization process) |
0401100000, 0401200000 |
Note: The above HS codes are the relevant HS codes for dairy products which require quarantine licenses, but it does not mean all products under these HS codes need quarantine licenses. Whether the product requires quarantine license or not will also depend on the production process.
Is your product required?
Only modified milk with pasteurization process under HS CODE 0401100000, 0401200000 will be required to obtain quarantine license. Otherwise, it is not required to obtain quarantine license.
Who?
The applicant who applies for quarantine license should be an independent legal entity in China that signs trade contract or agreement with overseas companies directly. Usually, the applicant is the importer.
When?
The applicant should apply for the quarantine license before signing the trade contract or agreement.
How?
Step 1: Apply for “filing of companies applying for quarantine license” and get the digital certificate (UKey).
Note: The period of validity for quarantine license is 6 months. The applicant should re-apply while exceeding the validity of the license or changing the product categories.
Step 2: Submit “Application form for entry animal and plant quarantine license” on “Review and Management System for Entry Animal and Plant Quarantine” online.
Step 3: Local Customs will conduct preliminary review. If passed, local Customs will submit their comments to General Administration of Customs for approval.
Step 4: General Administration of Customs will review and decide whether to approve the application or not.
Step 5: If approved, the applicant will receive the electronic version of quarantine license and can print it out for Customs inspection.
Note: The period of validity for quarantine license is 6 months. The applicant should re-apply while exceeding the validity of the license or changing the product categories.
Pasteurized milk under HS CODE 0401100000, 0401200000 will be required to obtain quarantine license.
Who?
The applicant who applies for quarantine license should be an independent legal entity in China that signs trade contract or agreement with overseas companies directly. Usually, the applicant is the importer.
When?
The applicant should apply for the quarantine license before signing the trade contract or agreement.
How?
Step 1: Apply for “filing of companies applying for quarantine license” and get the digital certificate (UKey).
Note: The period of validity for quarantine license is 6 months. The applicant should re-apply while exceeding the validity of the license or changing the product categories.
Step 2: Submit “Application form for entry animal and plant quarantine license” on “Review and Management System for Entry Animal and Plant Quarantine” online.
Step 3: Local Customs will conduct preliminary review. If passed, local Customs will submit their comments to General Administration of Customs for approval.
Step 4: General Administration of Customs will review and decide whether to approve the application or not.
Step 5: If approved, the applicant will receive the electronic version of quarantine license and can print it out for Customs inspection.
Note: The period of validity for quarantine license is 6 months. The applicant should re-apply while exceeding the validity of the license or changing the product categories.
Sterilized milk is not required to obtain quarantine license due to the production process.
Test Report
For dairy products, test report of the product shall be provided for the customs inspection to demonstrate that products comply with China’s food safety standards (GB standards). The test items are different for different situation. For first-time import (hereafter FI) product, the test results should be provided against all criteria in the GB standard. For subsequent import product (hereafter SI) , only part testing indexes required by AQSIQ are needed. But the copy of initial test report and inspection application form from FI should also be attached.
According to AQSIQ 2013 Announcement No. 53, first-time import (FI) means that the same product is imported to China through the same China port for the first time, and the same product means product with the same manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency. The follow-up import of the same product through the same port will be regarded as subsequent import. However, if any of the above information changed, say the China port or the product formula changed, the follow-up import will also be defined as FI.
Let’s take the following example for your better understanding. Product A has been imported to China through China port B with manufacturer C, product name D, trademark E, formula F, overseas exporter G and China’s importer/distributor/agency H for the first time. The follow-up import of product A will be regarded as FI or SI under different situation. Please refer to the table below for more details:
| Port | Manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency | FI or SI | |||||
| B | C, D, E, F, G, H | SI | |||||
| Not B | C, D, E, F, G, H | FI | |||||
| B | Any of C, D, E, F, G, H changed | FI | |||||
| Not B | Any of C, D, E, F, G, H changed | FI | |||||
What are the qualifications of the laboratories?
The report can be issued by official laboratory, third-party testing agency or corporate laboratory outside China, or by testing institutions with the Certificate and Mark of Qualification Certification for Food Inspection Agencies (CMAF) in China.
Please note that if the test report is issued by official laboratory, third-party testing agency or corporate laboratory outside China, a Chinese translation of the report should also be provided. And the test results should be converted to the units stipulated by the GB Standards.
List of testing items for FI and SI milk-based powder infant formula
The table below summarized the required testing items and their limits for both first-time import (FI) and subsequent import (SI) milk-based powder infant formula. ‘√’ means the corresponding index is requested, ‘×’ means the opposite. Please note that the testing of optional items are only required when those ingredients are labled on the package.
| Essential components | Items | FI | SI | Limits | ||||
| Per 100 kJ | Per 100 kcal | |||||||
| Minimum | Maximum | Minimum | Maximum | |||||
| Protein/(g) | √ | √ | 0.45 | 0.70 | 1.88 | 2.93 | The content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | √ | √ | 1.05 | 1.40 | 4.39 | 5.86 | ||
| In which: linoleic acid(g) | √ | × | 0.07 | 0.33 | 0.29 | 1.38 | ||
| α- linolenic acid (mg) | √ | × | 12 | N.S. | 50 | N.S. | N.S.: No specification | |
| Linoleic acid/α- linolenic acid ratio | √ | × | 5:1 | 15:1 | 5:1 | 15:1 | ||
| Erucic acid | √ | × | ≤1% of the total fatty acid | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Lauric acid and myristic acid | √ | × | ≤20% of the total fatty acid | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Total fatty acid | √ | × | N.S. | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Trans fatty acid | √ | × | ≤3% of the total fatty acid | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Total carbohydrate /(g) | √ | × | ≤5.0 | Carbohydrate content A1 should be calculated according to formula(1): A1= 100 − (A2+ A3+ A4+ A5+ A6) ………………………………(1) Where: A1——carbohydrate content, g/100g; A2——protein content, g/100g; A3——fat content, g/100g; A4——moisture content, g/100g; A5——ash content, g/100g; A6——dietary fiber content, g/100g. |
||||
| Lactose | √ | × | ≥90% of the total carbohydrate | For calculation of the proportion of lactose among the total carbohydrates, the added oligosaccharides and polysaccharides should not be included. | ||||
| Energy | √ | × | Within the range of 250 kJ (60 kcal)-295 kJ (70 kcal) per 100 mL in the ready-to-eat infant formulas . | The calculation of energy should be the value of the product of the content of protein, fat and carbohydrate per 100 mL product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber should be based on 50% of carbohydrate energy coefficient) respectively, and the sum (kJ/100mL) is divided by 4.184 to kcal/100 mL. | ||||
| Vitamin A /(μg RE) | √ | √ | 14 | 43 | 59 | 180 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | √ | √ | 0.25 | 0.60 | 1.05 | 2.51 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | √ | √ | 0.12 | 1.20 | 0.50 | 5.02 | 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3). | |
| Vitamin K1/(μg) | √ | √ | 1.0 | 6.5 | 4.2 | 27.2 | ||
| Vitamin B1/(μg) | √ | √ | 14 | 72 | 59 | 301 | ||
| Vitamin B2/(μg) | √ | √ | 19 | 119 | 80 | 498 | ||
| Vitamin B6/(μg) | √ | √ | 8.5 | 45.0 | 35.6 | 188.3 | ||
| Vitamin B12/(μg) | √ | √ | 0.025 | 0.360 | 0.105 | 1.506 | ||
| Niacin (niacinamide)/(μg) | √ | × | 70 | 360 | 293 | 1506 | Niacin: excludes precursor form. | |
| Folic acid/(μg) | √ | √ | 2.5 | 12.0 | 10.5 | 50.2 | ||
| Pantothenic acid/(μg) | √ | √ | 96 | 478 | 402 | 2000 | ||
| Vitamin C /(mg) | √ | × | 2.5 | 17.0 | 10.5 | 71.1 | ||
| Biotin/(μg) | √ | √ | 0.4 | 2.4 | 1.5 | 10.0 | ||
| Sodium/(mg) | √ | × | 5 | 14 | 21 | 59 | ||
| Potassium/(mg) | √ | × | 14 | 43 | 59 | 180 | ||
| Copper/(μg) | √ | √ | 8.5 | 29 | 35.6 | 121.3 | ||
| Magnesium/(mg) | √ | × | 1.2 | 3.6 | 5 | 15.1 | ||
| Iron/(mg) | √ | √ | 0.1 | 0.36 | 0.42 | 1.51 | ||
| Zinc/(mg) | √ | √ | 0.12 | 0.36 | 0.5 | 1.51 | ||
| Manganese/(mg) | √ | × | 1.2 | 24 | 5 | 100.4 | ||
| Calcium/(mg) | √ | √ | 12 | 35 | 50 | 146 | ||
| Phosphorus/(mg) | √ | √ | 6 | 24 | 25 | 100 | ||
| Calcium/phosphorus ratio | √ | × | 1:1 | 2:1 | 1:1 | 2:1 | ||
| Iodine/(μg) | √ | √ | 2.5 | 14 | 10.5 | 58.6 | ||
| Chloride/(mg) | √ | × | 12 | 38 | 50 | 159 | ||
| Selenium/(μg) | √ | √ | 0.48 | 1.9 | 2.01 | 7.95 | ||
| Optional components | Choline(mg) | √ | √ | 1.7 | 12 | 7.1 | 50.2 | |
| Inositol(mg) | √ | √ | 1 | 9.5 | 4.2 | 39.7 | ||
| Taurine(mg) | √ | √ | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine(mg) | √ | √ | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid (% total fatty acid) | √ | × | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) If docosahexaenoic acid (22:6 n-3) is supplemented to the infant formula, at least the same amount of Arachidonic acid (20:4 n-6) should be supplemented. Eicosapentaenoic acid (20:5 n-3) may exist in long chain unsaturated fatty acids, of which the total content should not exceed that of docosahexaenoic acid. 3) Total fatty acid refers to the sum of C4 ~ C24 fatty acids. |
|
| Arachidonic acid (% total fatty acid) | √ | × | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Moisture /(%) | √ | × | ≤5.0 | ||||
| Ash /(%) | √ | × | ≤4.0 | |||||
| Impurities /(mg/kg) | √ | × | ≤12 | |||||
| Limits of contaminants | Lead (Pb) /(mg/kg) | √ | √ | ≤0.15 | ||||
| Nitrate (based on NaNO3) /(mg/kg) | √ | √ | ≤100 | |||||
| Nitrite (based on NaNO2) /(mg/kg) | √ | √ | ≤2 | |||||
| Tin (Sn) /(mg/kg) | √ | × | ≤50 | Only limited to food packaged in tin plate containers. | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | √ | √ | ≤0.5 | ||||
| Limits of microorganisms | Items | FI | SI | Limits (if not otherwise specified, in CFU/g(ml)) | n: total sample number in 1 batch c: maximum tolerance number of sample, of which testing result between m and M. m: microbiological acceptance level M: microbiological maximum safety tolerance |
|||
| n | c | m | M | |||||
| Aerobic plate count | √ | √ | 5 | 2 | 1000 | 10000 | Not applicable to products supplemented with active bacteria (aerobic and anaerobic probiotics) [the number of active bacteria of active probiotics in products should be ≥106 CFU/g(ml)]. | |
| Coliform bacteria | √ | √ | 5 | 2 | 10 | 100 | ||
| Staphylococcus aureus | √ | √ | 5 | 2 | 10 | 100 | ||
| Enterobacter sakazakii | √ | √ | 3 | 0 | 0/100g | – | Only applicable to formulas for infants of 0-6 months old. | |
| Salmonella | √ | √ | 5 | 0 | 0/25g | – | ||
| Urease activity | Qualitative determination of urease activity | √ | × | Negative | Only applicable to products containing soy component | |||
According to AQSIQ 2013 Announcement No. 53, first-time import (FI) means that the same product is imported to China through the same China port for the first time, and the same product means product with the same manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency. The follow-up import of the same product through the same port will be regarded as subsequent import. However, if any of the above information changed, say the China port or the product formula changed, the follow-up import will also be defined as FI.
Let’s take the following example for your better understanding. Product A has been imported to China through China port B with manufacturer C, product name D, trademark E, formula F, overseas exporter G and China’s importer/distributor/agency H for the first time. The follow-up import of product A will be regarded as FI or SI under different situation. Please refer to the table below for more details:
| Port | Manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency | FI or SI | |||||
| B | C, D, E, F, G, H | SI | |||||
| Not B | C, D, E, F, G, H | FI | |||||
| B | Any of C, D, E, F, G, H changed | FI | |||||
| Not B | Any of C, D, E, F, G, H changed | FI | |||||
What are the qualifications of the laboratories?
The report can be issued by official laboratory, third-party testing agency or corporate laboratory outside China, or by testing institutions with the Certificate and Mark of Qualification Certification for Food Inspection Agencies (CMAF) in China.
Please note that if the test report is issued by official laboratory, third-party testing agency or corporate laboratory outside China, a Chinese translation of the report should also be provided. And the test results should be converted to the units stipulated by the Chinese natinoal standards. ”
List of testing items for FI and SI milk-based powder infant formula
The table below summarized the required testing items and their limits for both first-time import (FI) and subsequent import (SI) milk-based liquid infant formula. ‘√’ means the corresponding index is requested, ‘×’ means the opposite. Please note that the testing of optional items are only required when those ingredients are labled on the package.
| Essential components | Items | FI | SI | Limits | ||||
| Per 100 kJ | Per 100 kcal | |||||||
| Minimum | Maximum | Minimum | Maximum | |||||
| Protein/(g) | √ | √ | 0.45 | 0.70 | 1.88 | 2.93 | The content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | √ | √ | 1.05 | 1.40 | 4.39 | 5.86 | ||
| In which: linoleic acid(g) | √ | × | 0.07 | 0.33 | 0.29 | 1.38 | ||
| α- linolenic acid (mg) | √ | × | 12 | N.S. | 50 | N.S. | N.S.: No specification | |
| Linoleic acid/α- linolenic acid ratio | √ | × | 5:1 | 15:1 | 5:1 | 15:1 | ||
| Erucic acid | √ | × | ≤1% of the total fatty acid | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Lauric acid and myristic acid | √ | × | ≤20% of the total fatty acid | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Total fatty acid | √ | × | N.S. | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Trans fatty acid | √ | × | ≤3% of the total fatty acid | The total fatty acid refers to the sum of C4~C24 fatty acid. | ||||
| Total carbohydrate /(g) | √ | × | ≤5.0 | Carbohydrate content A1 should be calculated according to formula(1): A1= 100 − (A2+ A3+ A4+ A5+ A6) ………………………………(1) Where: A1——carbohydrate content, g/100g; A2——protein content, g/100g; A3——fat content, g/100g; A4——moisture content, g/100g; A5——ash content, g/100g; A6——dietary fiber content, g/100g. |
||||
| Lactose | √ | × | ≥90% of the total carbohydrate | For calculation of the proportion of lactose among the total carbohydrates, the added oligosaccharides and polysaccharides should not be included. | ||||
| Energy | √ | × | Within the range of 250 kJ (60 kcal)-295 kJ (70 kcal) per 100 mL in the ready-to-eat infant formulas . | The calculation of energy should be the value of the product of the content of protein, fat and carbohydrate per 100 mL product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber should be based on 50% of carbohydrate energy coefficient) respectively, and the sum (kJ/100mL) is divided by 4.184 to kcal/100 mL. | ||||
| Vitamin A /(μg RE) | √ | √ | 14 | 43 | 59 | 180 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | √ | √ | 0.25 | 0.60 | 1.05 | 2.51 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | √ | √ | 0.12 | 1.20 | 0.50 | 5.02 | 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3). | |
| Vitamin K1/(μg) | √ | √ | 1.0 | 6.5 | 4.2 | 27.2 | ||
| Vitamin B1/(μg) | √ | √ | 14 | 72 | 59 | 301 | ||
| Vitamin B2/(μg) | √ | √ | 19 | 119 | 80 | 498 | ||
| Vitamin B6/(μg) | √ | √ | 8.5 | 45.0 | 35.6 | 188.3 | ||
| Vitamin B12/(μg) | √ | √ | 0.025 | 0.360 | 0.105 | 1.506 | ||
| Niacin (niacinamide)/(μg) | √ | × | 70 | 360 | 293 | 1506 | Niacin: excludes precursor form. | |
| Folic acid/(μg) | √ | √ | 2.5 | 12.0 | 10.5 | 50.2 | ||
| Pantothenic acid/(μg) | √ | √ | 96 | 478 | 402 | 2000 | ||
| Vitamin C /(mg) | √ | × | 2.5 | 17.0 | 10.5 | 71.1 | ||
| Biotin/(μg) | √ | √ | 0.4 | 2.4 | 1.5 | 10.0 | ||
| Sodium/(mg) | √ | × | 5 | 14 | 21 | 59 | ||
| Potassium/(mg) | √ | × | 14 | 43 | 59 | 180 | ||
| Copper/(μg) | √ | √ | 8.5 | 29 | 35.6 | 121.3 | ||
| Magnesium/(mg) | √ | × | 1.2 | 3.6 | 5 | 15.1 | ||
| Iron/(mg) | √ | √ | 0.1 | 0.36 | 0.42 | 1.51 | ||
| Zinc/(mg) | √ | √ | 0.12 | 0.36 | 0.5 | 1.51 | ||
| Manganese/(mg) | √ | × | 1.2 | 24 | 5 | 100.4 | ||
| Calcium/(mg) | √ | √ | 12 | 35 | 50 | 146 | ||
| Phosphorus/(mg) | √ | √ | 6 | 24 | 25 | 100 | ||
| Calcium/phosphorus ratio | √ | × | 1:1 | 2:1 | 1:1 | 2:1 | ||
| Iodine/(μg) | √ | √ | 2.5 | 14 | 10.5 | 58.6 | ||
| Chloride/(mg) | √ | × | 12 | 38 | 50 | 159 | ||
| Selenium/(μg) | √ | √ | 0.48 | 1.9 | 2.01 | 7.95 | ||
| Optional components | Choline(mg) | √ | √ | 1.7 | 12 | 7.1 | 50.2 | |
| Inositol(mg) | √ | √ | 1 | 9.5 | 4.2 | 39.7 | ||
| Taurine(mg) | √ | √ | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine(mg) | √ | √ | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid (% total fatty acid) | √ | × | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) If docosahexaenoic acid (22:6 n-3) is supplemented to the infant formula, at least the same amount of Arachidonic acid (20:4 n-6) should be supplemented. Eicosapentaenoic acid (20:5 n-3) may exist in long chain unsaturated fatty acids, of which the total content should not exceed that of docosahexaenoic acid. 3) Total fatty acid refers to the sum of C4 ~ C24 fatty acids. |
|
| Arachidonic acid (% total fatty acid) | √ | × | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Moisture /(%) | √ | × | ≤5.0 | ||||
| Ash (calculated by dry substance) /(%) | √ | × | ≤4.2 | |||||
| Impurities /(mg/kg) | √ | × | ≤2 | |||||
| Limits of contaminants | Lead (Pb) /(mg/kg) | √ | √ | ≤0.15 | ||||
| Nitrate (based on NaNO3) /(mg/kg) | √ | √ | ≤100 | |||||
| Nitrite (based on NaNO2) /(mg/kg) | √ | √ | ≤2 | |||||
| Tin (Sn) /(mg/kg) | √ | × | ≤50 | Only limited to food packaged in tin plate containers. | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | √ | √ | ≤0.5 | ||||
| Limits of microorganisms | Limits of microorganisms | √ | √ | commercial sterilization | ||||
| Urease activity | Qualitative determination of urease activity | √ | × | Negative | 1) Only applicable to products containing soy component; 2) The sampling quantity of liquid infant formulas should be converted according to the content of dry substances. |
|||
According to AQSIQ 2013 Announcement No. 53, first-time import (FI) means that the same product is imported to China through the same China port for the first time, and the same product means product with the same manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency. The follow-up import of the same product through the same port will be regarded as subsequent import. However, if any of the above information changed, say the China port or the product formula changed, the follow-up import will also be defined as FI.
Let’s take the following example for your better understanding. Product A has been imported to China through China port B with manufacturer C, product name D, trademark E, formula F, overseas exporter G and China’s importer/distributor/agency H for the first time. The follow-up import of product A will be regarded as FI or SI under different situation. Please refer to the table below for more details:
| Port | Manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency | FI or SI | |||||
| B | C, D, E, F, G, H | SI | |||||
| Not B | C, D, E, F, G, H | FI | |||||
| B | Any of C, D, E, F, G, H changed | FI | |||||
| Not B | Any of C, D, E, F, G, H changed | FI | |||||
What are the qualifications of the laboratories?
The report can be issued by official laboratory, third-party testing agency or corporate laboratory outside China, or by testing institutions with the Certificate and Mark of Qualification Certification for Food Inspection Agencies (CMAF) in China.
Please note that if the test report is issued by official laboratory, third-party testing agency or corporate laboratory outside China, a Chinese translation of the report should also be provided. And the test results should be converted to the units stipulated by the Chinese natinoal standards.
List of testing items for FI and SI milk-based powder older infant formula and young children formula
The table below summarized the required testing items and their limits for both first-time import (FI) and subsequent import (SI) milk-based powder older infant formula and young children formula. ‘√’ means the corresponding index is requested, ‘×’ means the opposite. Please note that the testing of optional items are only required when those ingredients are labled on the package.
| Essential components | Items | FI | SI | Limits | ||||
| Per 100 kJ | Per 100 kcal | |||||||
| Minimum | Maximum | Minimum | Maximum | |||||
| Protein/(g) | √ | √ | 0.7 | 1.2 | 2.9 | 5.0 | The content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | √ | √ | 0.7 | 1.4 | 2.9 | 5.9 | ||
| In which: linoleic acid(g) | √ | × | 0.07 | N.S. | 0.29 | N.S. | ||
| Total fatty acid | √ | × | N.S. | 1) The total fatty acid refers to the sum of C4~C24 fatty acid; 2) N.S.: No specification. | ||||
| Trans fatty acid | √ | × | ≤3% of the total fatty acid | |||||
| Carbohydrate /(g) | √ | × | N.S. | N.S.: No specification | ||||
| Energy | √ | × | Within the range of 250 kJ (60 kcal)-355 kJ (85 kcal) per 100 mL in the ready-to-eat older infants and young children formula. | For calculation of energy, the content of protein, fat, carbohydrate per 100mL of product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber is calculated as 50% of carbohydrate energy coefficient) respectively, the obtained sum (kJ/100mL) divided by 4.184 to obtain the kcal/100mL value. | ||||
| Vitamin A /(μg RE) | √ | √ | 18 | 54 | 75 | 225 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | √ | √ | 0.25 | 0.75 | 1.05 | 3.14 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | √ | √ | 0.15 | N.S. | 0.63 | N.S. | 1) 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3); 2) N.S.: No specification |
|
| Vitamin K1/(μg) | √ | √ | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Vitamin B1/(μg) | √ | √ | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B2/(μg) | √ | √ | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B6/(μg) | √ | √ | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B12/(μg) | √ | √ | 0.04 | N.S. | 0.17 | N.S. | N.S.: No specification | |
| Niacin (niacinamide)/(μg) | √ | × | 110 | N.S. | 460 | N.S. | 1) Niacin: excludes precursor form; 2) N.S.: No specification | |
| Folic acid/(μg) | √ | √ | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Pantothenic acid/(μg) | √ | √ | 70 | N.S. | 293 | N.S. | N.S.: No specification | |
| Vitamin C /(mg) | √ | × | 1.8 | N.S. | 7.5 | N.S. | N.S.: No specification | |
| Biotin/(μg) | √ | √ | 0.4 | N.S. | 1.7 | N.S. | N.S.: No specification | |
| Sodium/(mg) | √ | × | N.S. | 20 | N.S. | 84 | N.S.: No specification | |
| Potassium/(mg) | √ | × | 18 | 69 | 75 | 289 | ||
| Copper/(μg) | √ | √ | 7 | 35 | 29 | 146 | ||
| Magnesium/(mg) | √ | × | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Iron/(mg) | √ | √ | 0.25 | 0.5 | 1.05 | 2.09 | ||
| Zinc/(mg) | √ | √ | 0.1 | 0.3 | 0.4 | 1.3 | ||
| Calcium/(mg) | √ | √ | 17 | N.S. | 71 | N.S. | N.S.: No specification | |
| Phosphorus/(mg) | √ | √ | 8.3 | N.S. | 34.7 | N.S. | N.S.: No specification | |
| Calcium/phosphorus ratio | √ | × | 1.2:1 | 2:1 | 1.2:1 | 2:1 | ||
| Iodine/(μg) | √ | √ | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Chloride/(mg) | √ | × | N.S. | 52 | N.S. | 218 | N.S.: No specification | |
| Optional components | Selenium /(μg) | √ | √ | 0.48 | 1.90 | 2.01 | 7.95 | |
| Choline /(mg) | √ | √ | 1.7 | 12.0 | 7.1 | 50.2 | ||
| Manganese /(μg) | √ | × | 0.25 | 24.0 | 1.05 | 100.4 | ||
| Inositol /(mg) | √ | √ | 1.0 | 9.5 | 4.2 | 39.7 | ||
| Taurine /(mg) | √ | √ | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine /(mg) | √ | √ | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid /(% total fatty acid) | √ | × | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) Total fatty acid refers to the sum of C4-C24 fatty acids. |
|
| Arachidonic acid /(% total fatty acid) | √ | × | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Moisture /(%) | √ | × | ≤5.0 | ||||
| Ash /(%) | √ | × | ≤5.0 | |||||
| Impurities /(mg/kg) | √ | × | ≤12 | NOT applicable to products supplemented with fruits and vegetables. | ||||
| Limits of contaminants | Lead (Pb) /(mg/kg) | √ | √ | ≤0.15 | ||||
| Nitrate (based on NaNO3) /(mg/kg) | √ | √ | ≤100 | NOT applicable to products supplemented with fruits and vegetables | ||||
| Nitrite (based on NaNO2) /(mg/kg) | √ | √ | ≤2.0 | |||||
| Tin (Sn) /(mg/kg) | √ | × | ≤50 | Only limited to food packaged in tin plate containers. | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | √ | √ | ≤0.5 | ||||
| Limits of microorganisms | Items | FI | SI | Limits (if not otherwise specified, in CFU/g(ml)) | n: total sample number in 1 batch c: maximum tolerance number of sample, of which testing result between m and M. m: microbiological acceptance level M: microbiological maximum safety tolerance |
|||
| n | c | m | M | |||||
| Aerobic plate count | √ | √ | 5 | 2 | 1000 | 10000 | Not applicable to products supplemented with active bacteria (aerobic and anaerobic probiotics) [the number of active bacteria of active probiotics in products should be ≥106 CFU/g(ml)]. | |
| Coliform bacteria | √ | √ | 5 | 2 | 10 | 100 | ||
| Salmonella | √ | √ | 5 | 0 | 0/25g | – | ||
| Urease activity | Qualitative determination of urease activity | √ | × | Negative | Only applicable to products containing soy component | |||
Essential componentsItemsFISILimits
Per 100 kJPer 100 kcal
MinimumMaximumMinimumMaximum
Protein/(g)√√0.450.701.882.93The content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25Fat/(g)√√1.051.404.395.86
In which: linoleic acid(g)√×0.070.330.291.38
α- linolenic acid (mg)√×12N.S.50N.S.N.S.: No specification Linoleic acid/α- linolenic acid ratio√×5:115:15:115:1
Erucic acid √×≤1% of the total fatty acidThe total fatty acid refers to the sum of C4~C24 fatty acid. Lauric acid and myristic acid √×≤20% of the total fatty acidThe total fatty acid refers to the sum of C4~C24 fatty acid. Total fatty acid√×N.S.The total fatty acid refers to the sum of C4~C24 fatty acid. Trans fatty acid √×≤3% of the total fatty acidThe total fatty acid refers to the sum of C4~C24 fatty acid.Total carbohydrate /(g)√×≤5.0Carbohydrate content A1 should be calculated according to formula(1):
A1= 100 − (A2+ A3+ A4+ A5+ A6) ………………………………(1)
Where:
A1——carbohydrate content, g/100g;
A2——protein content, g/100g;
A3——fat content, g/100g;
A4——moisture content, g/100g;
A5——ash content, g/100g;
A6——dietary fiber content, g/100g. Lactose√× ≥90% of the total carbohydrateFor calculation of the proportion of lactose among the total carbohydrates, the added oligosaccharides and polysaccharides should not be included.Energy√×Within the range of 250 kJ (60 kcal)-295 kJ (70 kcal) per 100 mL in the ready-to-eat infant formulas . The calculation of energy should be the value of the product of the content of protein, fat and carbohydrate per 100 mL product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber should be based on 50% of carbohydrate energy coefficient) respectively, and the sum (kJ/100mL) is divided by 4.184 to kcal/100 mL.Vitamin A /(μg RE)√√144359180RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A.Vitamin D /(μg)√√0.250.601.052.51Calciferol, 1μg Vitamin D = 40 IU Vitamin D Vitamin E /(mg α-TE)√√0.121.200.505.021 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3).Vitamin K1/(μg)√√1.06.54.227.2
Vitamin B1/(μg)√√147259301
Vitamin B2/(μg)√√1911980498
Vitamin B6/(μg)√√8.545.035.6188.3
Vitamin B12/(μg)√√0.0250.3600.1051.506
Niacin (niacinamide)/(μg)√×703602931506Niacin: excludes precursor form.Folic acid/(μg)√√2.512.010.550.2
Pantothenic acid/(μg)√√964784022000
Vitamin C /(mg)√×2.517.010.571.1
Biotin/(μg)√√0.42.41.510.0
Sodium/(mg)√×5142159
Potassium/(mg)√×144359180
Copper/(μg)√√8.52935.6121.3
Magnesium/(mg)√×1.23.6515.1
Iron/(mg)√√0.10.360.421.51
Zinc/(mg)√√0.120.360.51.51
Manganese/(mg)√×1.2245100.4
Calcium/(mg)√√123550146
Phosphorus/(mg)√√62425100
Calcium/phosphorus ratio√×1:12:11:12:1
Iodine/(μg)√√2.51410.558.6
Chloride/(mg)√×123850159
Selenium/(μg)√√0.481.92.017.95
Optional componentsCholine(mg)√√1.7127.150.2
Inositol(mg)√√19.54.239.7
Taurine(mg)√√N.S.3N.S.13N.S.: No specificationL-Carnitine(mg)√√0.3N.S.1.3N.S.N.S.: No specificationDocosahexaenoic acid (% total fatty acid)√×N.S.0.5N.S.0.51) N.S.: No specification;
2) If docosahexaenoic acid (22:6 n-3) is supplemented to the infant formula, at least the same amount of Arachidonic acid (20:4 n-6) should be supplemented. Eicosapentaenoic acid (20:5 n-3) may exist in long chain unsaturated fatty acids, of which the total content should not exceed that of docosahexaenoic acid.
3) Total fatty acid refers to the sum of C4 ~ C24 fatty acids.Arachidonic acid (% total fatty acid)√×N.S.1N.S.1Other indexesMoisture /(%)√×≤5.0
Ash (calculated by dry substance) /(%)√×≤4.2
Impurities /(mg/kg)√×≤2
Limits of contaminantsLead (Pb) /(mg/kg) √√≤0.15
Nitrate (based on NaNO3) /(mg/kg) √√≤100
Nitrite (based on NaNO2) /(mg/kg)√√≤2
Tin (Sn) /(mg/kg)√×≤50Only limited to food packaged in tin plate containers.Limits of mycotoxinsAflatoxin M1/(μg/kg) √√≤0.5
Limits of microorganismsLimits of microorganisms√√commercial sterilization
Urease activityQualitative determination of urease activity√×Negative1) Only applicable to products containing soy component;
2) The sampling quantity of liquid infant formulas should be converted according to the content of dry substances.
According to AQSIQ 2013 Announcement No. 53, first-time import (FI) means that the same product is imported to China through the same China port for the first time, and the same product means product with the same manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency. The follow-up import of the same product through the same port will be regarded as subsequent import. However, if any of the above information changed, say the China port or the product formula changed, the follow-up import will also be defined as FI.
Let’s take the following example for your better understanding. Product A has been imported to China through China port B with manufacturer C, product name D, trademark E, formula F, overseas exporter G and China’s importer/distributor/agency H for the first time. The follow-up import of product A will be regarded as FI or SI under different situation. Please refer to the table below for more details:
| Port | Manufacturer, product name (includes trademark), formula, overseas exporter, China’s importer/distributor/agency | FI or SI | |||||
| B | C, D, E, F, G, H | SI | |||||
| Not B | C, D, E, F, G, H | FI | |||||
| B | Any of C, D, E, F, G, H changed | FI | |||||
| Not B | Any of C, D, E, F, G, H changed | FI | |||||
What are the qualifications of the laboratories?
The report can be issued by official laboratory, third-party testing agency or corporate laboratory outside China, or by testing institutions with the Certificate and Mark of Qualification Certification for Food Inspection Agencies (CMAF) in China.
Please note that if the test report is issued by official laboratory, third-party testing agency or corporate laboratory outside China, a Chinese translation of the report should also be provided. And the test results should be converted to the units stipulated by the Chinese natinoal standards.
List of testing items for FI and SI milk-based liquid older infant formula and young children formula
The table below summarized the required testing items and their limits for both first-time import (FI) and subsequent import (SI) milk-based powder older infant formula and young children formula. ‘√’ means the corresponding index is requested, ‘×’ means the opposite. Please note that the testing of optional items are only required when those ingredients are labled on the package.
| Essential components | Items | FI | SI | Limits | ||||
| Per 100 kJ | Per 100 kcal | |||||||
| Minimum | Maximum | Minimum | Maximum | |||||
| Protein/(g) | √ | √ | 0.7 | 1.2 | 2.9 | 5.0 | The content of whey protein should be over or equal to 60%; the content of protein should be calculated as nitrogen (N) × 6.25 | |
| Fat/(g) | √ | √ | 0.7 | 1.4 | 2.9 | 5.9 | ||
| In which: linoleic acid(g) | √ | × | 0.07 | N.S. | 0.29 | N.S. | ||
| Total fatty acid | √ | × | N.S. | 1) The total fatty acid refers to the sum of C4~C24 fatty acid; 2) N.S.: No specification. | ||||
| Trans fatty acid | √ | × | ≤3% of the total fatty acid | |||||
| Carbohydrate /(g) | √ | × | N.S. | N.S.: No specification | ||||
| Energy | √ | × | Within the range of 250 kJ (60 kcal)-355 kJ (85 kcal) per 100 mL in the ready-to-eat older infants and young children formula. | For calculation of energy, the content of protein, fat, carbohydrate per 100mL of product multiplied by the energy coefficient of 17 kJ/g, 37 kJ/g, 17 kJ/g (the energy coefficient of dietary fiber is calculated as 50% of carbohydrate energy coefficient) respectively, the obtained sum (kJ/100mL) divided by 4.184 to obtain the kcal/100mL value. | ||||
| Vitamin A /(μg RE) | √ | √ | 18 | 54 | 75 | 225 | RE is retinol equivalent. 1μg RE=1μg All trans retinol (Vitamin A) =3.33 IU Vitamin A. Ingredients of Vitamin A shall only include preformed retinol. No carotenoids ingredient shall be included when calculating or claiming activities of Vitamin A. | |
| Vitamin D /(μg) | √ | √ | 0.25 | 0.75 | 1.05 | 3.14 | Calciferol, 1μg Vitamin D = 40 IU Vitamin D | |
| Vitamin E /(mg α-TE) | √ | √ | 0.15 | N.S. | 0.63 | N.S. | 1) 1 mg α-TE (α-tocopherol equivalent) =1 mg d-α-tocopherol. The content of Vitamin E should be at least 0.5mg of α-TE per gram of polyunsaturated fatty acid. The minimum content of Vitamin E content should be regulated according to the number of double bonds in polyunsaturated fatty acids in the formula as follows: 0.5 mg of α-TE per gram of linoleic acid (18:2 n-6); 0.75 mg ofα-TE per gram ofα-linolenic acid (18:3 n-3); 1.0 mg ofα-TE per gram of arachidonic acid (20:4 n-6); 1.25mg of α-TE per gram of Eicosapentaenoic Acid (20:5 n-3); 1.5mg of α-TE per gram of docosahexenoic acid (22:6 n-3); 2) N.S.: No specification |
|
| Vitamin K1/(μg) | √ | √ | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Vitamin B1/(μg) | √ | √ | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B2/(μg) | √ | √ | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B6/(μg) | √ | √ | 11 | N.S. | 46 | N.S. | N.S.: No specification | |
| Vitamin B12/(μg) | √ | √ | 0.04 | N.S. | 0.17 | N.S. | N.S.: No specification | |
| Niacin (niacinamide)/(μg) | √ | × | 110 | N.S. | 460 | N.S. | 1) Niacin: excludes precursor form; 2) N.S.: No specification | |
| Folic acid/(μg) | √ | √ | 1 | N.S. | 4 | N.S. | N.S.: No specification | |
| Pantothenic acid/(μg) | √ | √ | 70 | N.S. | 293 | N.S. | N.S.: No specification | |
| Vitamin C /(mg) | √ | × | 1.8 | N.S. | 7.5 | N.S. | N.S.: No specification | |
| Biotin/(μg) | √ | √ | 0.4 | N.S. | 1.7 | N.S. | N.S.: No specification | |
| Sodium/(mg) | √ | × | N.S. | 20 | N.S. | 84 | N.S.: No specification | |
| Potassium/(mg) | √ | × | 18 | 69 | 75 | 289 | ||
| Copper/(μg) | √ | √ | 7 | 35 | 29 | 146 | ||
| Magnesium/(mg) | √ | × | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Iron/(mg) | √ | √ | 0.25 | 0.5 | 1.05 | 2.09 | ||
| Zinc/(mg) | √ | √ | 0.1 | 0.3 | 0.4 | 1.3 | ||
| Calcium/(mg) | √ | √ | 17 | N.S. | 71 | N.S. | N.S.: No specification | |
| Phosphorus/(mg) | √ | √ | 8.3 | N.S. | 34.7 | N.S. | N.S.: No specification | |
| Calcium/phosphorus ratio | √ | × | 1.2:1 | 2:1 | 1.2:1 | 2:1 | ||
| Iodine/(μg) | √ | √ | 1.4 | N.S. | 5.9 | N.S. | N.S.: No specification | |
| Chloride/(mg) | √ | × | N.S. | 52 | N.S. | 218 | N.S.: No specification | |
| Optional components | Selenium /(μg) | √ | √ | 0.48 | 1.90 | 2.01 | 7.95 | |
| Choline /(mg) | √ | √ | 1.7 | 12.0 | 7.1 | 50.2 | ||
| Manganese /(μg) | √ | × | 0.25 | 24.0 | 1.05 | 100.4 | ||
| Inositol /(mg) | √ | √ | 1.0 | 9.5 | 4.2 | 39.7 | ||
| Taurine /(mg) | √ | √ | N.S. | 3 | N.S. | 13 | N.S.: No specification | |
| L-Carnitine /(mg) | √ | √ | 0.3 | N.S. | 1.3 | N.S. | N.S.: No specification | |
| Docosahexaenoic acid /(% total fatty acid) | √ | × | N.S. | 0.5 | N.S. | 0.5 | 1) N.S.: No specification; 2) Total fatty acid refers to the sum of C4-C24 fatty acids. |
|
| Arachidonic acid /(% total fatty acid) | √ | × | N.S. | 1 | N.S. | 1 | ||
| Other indexes | Ash (calculated by dry substance) /(%) | √ | × | ≤5.3 | ||||
| Impurities /(mg/kg) | √ | × | ≤2 | NOT applicable to products supplemented with fruits and vegetables. | ||||
| Limits of contaminants | Lead (Pb) /(mg/kg) | √ | √ | ≤0.15 | ||||
| Nitrate (based on NaNO3) /(mg/kg) | √ | √ | ≤100 | as ready-to-usestate | ||||
| Nitrite (based on NaNO2) /(mg/kg) | √ | √ | ≤2.0 | 1) NOT applicable to products supplemented with fruits and vegetables; 2) calculated based on powder product. | ||||
| Tin (Sn) /(mg/kg) | √ | × | ≤50 | calculated based on powder product | ||||
| Limits of mycotoxins | Aflatoxin M1/(μg/kg) | √ | √ | ≤0.5 | Only limited to food packaged in tin plate containers. | |||
| Limits of microorganisms | Limits of microorganisms | √ | √ | commercial sterilization | calculated based on powder product | |||
| Urease activity | Qualitative determination of urease activity | √ | × | Negative | 1) Products containing soy component should be consistent with this provision; 2) The sampling quantity of liquid infant formulas should be converted according to the content of dry substances. |
|||
Customs Inspection Check
After product shipment arrives at China port, the products will be stored in a designated location (warehouse) before final clearance. The importer who has Customs declaration qualification (or entrusted professional Customs broker) will submit relevant documents for Customs inspection and Clearance. Then Customs will check the documents and conduct sampling test. If all documents and the test result are compliant, after paying the tariffs, the importer will get an Inspection and Quarantine Certificate for imported goods, and Customs will release the goods.
Usually, it will take about 5-20 working days for Customs inspection and clearance. It may be shortened or prolonged according to different factors, such as preparation of documents, importer credit rating, communication with Customs, etc.
This checklist is for Customs Inspection declaration. Customs will check those items according to product category and practical situation. Please refer to the following checklist for more details.
Check-list items
| No. | Check-list | Remark | |
| 1 | Automatic import license | 1) What is that? | Automatic import license refers to the license issued by MOC authoried agency to management the quantity or other restrictions in accordance with the regulations (see Pic.1). |
| Pic.1 |  |
||
| 2) Is your product required? | All dairy products need to get license of automatic import including fresh milk, whey and milk powder. | ||
| 3) If so, Who will obtain? | The Chinese importer need to apply the license before cargos arrived. | ||
| 2 | Certificate of origin (COO) | It is required for all dairy products. The exporter can obtain COO from those local accredited bodies.For more details on this section, please click here. |
|
| 3 | Foreign official health certificate confirmed by China government | 1) What is that? | Health certificate is issued by Australia government to confirm the product is produced in sound condition, fit for human consumption and of Australia origin (see Pic.2 HEALTH CERTIFICATE ). |
| Pic.2 |  |
||
| 2) Is your product required? | Yes, all dairy products need to get this certificate. | ||
| 3) If so, Who will obtain? | Exporter shall apply for it from Department of Agriculture and Water Resources. Please pay special attention to the following requirements: 1) The Health Certificate should be original with official seal and signature of official veterinarian; 2) The country of final destination should be the People’s Republic of China; 3) The form of the certificate and the official seal shall be the same as the version confirmed by China government; 4) The product information including name, specification, production batch, net weight or count, etc. on the certificate should be consistent with the declared product.For more details on this section, click here. |
||
| 4 | Certificate of wooden fumigation for wooden packing | Applicable to wooden packing. Importers in China need to apply the certificate with Customs. After getting the certificate, your product would be printed IPPC logo (see Pic.3). | |
| Pic.3 |  |
||
| 5 | Test report | 1) What is that? | Test report of the product shall be provided for the customs inspection to demonstrate that testing results of the products comply with China standards. |
| 2) Is your product required? | Yes, it is required for all dairy products. Test report of the product shall be provided to the customs official for review to demonstrate that products comply with the testing items and parameters in the China’s food safety standards (GB standards). | ||
| 3) If so, Who will obtain? | It can be provided either by exporter or by importer. | ||
| 4) If so, how to obtain? | 1) The report can be issued by the overseas official laboratory, third-party testing agency or the enterprise laboratory, or by testing institutions with the Certificate and Mark of Qualification Certification for Food Inspection Agencies (CMAF) in China. It is suggested to test with GB methods because the test results may be different due to different methods. 2) If the test report is not in Chinese, a corresponding Chinese translation should also be attached. 3) Please note that each batch should have a Certificate of Analysis (COA) and the test items required are more demanding for first-time import compared with subsequent imports. For more details on requirements for test reports, please click here. |
||
| 6 | Contract, invoice, packing list and bill of lading | In order to trace the origin of the product, those documents refers to the supporting documents of shipment. | |
| 7 | License for Quarantine of entry animals and plants | The categories of dairy products need to get license of quarantine including: Raw milk (HS code: 0401200000), Raw milk products (HS code: 0403100000, 0406100000, 0406200000, 0406300000, 0406400000, 0406900000); Pasteurized milk and pasteurized modified milk (HS code: 0401100000, 0401200000). Importers need to apply for the license before cargoes arrived. |
|
| 8 | The sample of original label and Chinese translation, the sample of Chinese label | 1) What is that? | Original label refers to the label of the product used in Australia. The Chinese translation refers to translation all information on the original label into simplified Chinese, with the exception of the English brand name, English name and address of manufacturer/exporter/agent outside China. The sample of Chinese label is the label intended to be applied on the product after imported into China, which must meet the relevant Chinese labelling laws, regulations and GB standards. |
| 2) Is your product required? | 1) For first-time imported dairy products, the sample of original label, Chinese translation and the sample of Chinese label shall be provided. If requirements are met, a record number of label will be issued. 2) For subsequent imported dairy products, the sample of Chinese label and the record number of label are required. |
||
| 3) If so, Who will obtain? | The manufacturer or exporter should provide the Chinese label. However, in most cases it would be complicated to design your own Chinese label so it is recommend to seek a qualified expert to design a Chinese label according to Chinese food labelling regulations. | ||
| 4) If so, what type of Chinese labelling? | The Chinese label can be printed directly on the packaging or a pasted Chinese label stuck over the original English label which can be stuck on before or after products arrives at China port in the specified warehouse in the bonded area (Often times this can be a cost effective method, which can be organised by the importer/logistics agent/customs broker when goods arrive at China port). For more details on Chinese labelling requirements, please click here. | ||
| 9 | Others | 1) Certificate of conformity for nutritional information | All products with nutritional facts should provide materials to demonstrate compliance. |
| 2) Copy of the business license of the Chinese importer/distributor/agent listed on the label | Only for first-time imported products. The Chinese importer/distributor/agent will prepare for it. | ||
| 3) Evidentiary documents for emphasized information on the labelling | If there’s some special information emphasized on the labelling of the product, such as prize, honour, legal producing area, geographical indication, certification logos,or special components; corresponding evidentiary documents shall be provided by the manufacturer/exporter/agent. Please note that organic labelling requires China organic certification. | ||
Suggestions
- Exporters should equip themselves with enough regulatory knowledge to ask the right questions regarding Chinese regulations and standards and are recommended to pay particular attention to continuously updated regulations.
- For first-time shipments, it is suggested to airfreight a small batch of products before exporting larger quantities.
- It is suggested to find a qualified and experienced importer who has a good relationship with Customs. You can learn more about how to find a good importer through the Customs Inspection Credit Management System located under ‘Value of Partnerships‘ in this section.
- Exporter should maintain a good relationship and communication with the importer and consult with expert advisors to support in China compliance and other areas for smoother market access
- Customs declaration is one of the most important step of product importation. Well prepared, dilligent and accurate documentatiom will reduce the risk of problems and speed up market access during inspection.
Note
Please note that the checklist provided is for reference only and may not be complete for all circumstances.
Customs Taxes
Tariff Rates
China Customs will assess and collect the tariffs (Tax). The import tariff rate can be divided into the following categories: General Rates, Most-Favored-Nation (MFN) rates, agreement rates such as ChAFTA, Preferential rates, Tariff Rate Quota Rates, and Provisional rates. Australia and China has signed the China Australia Free Trade Agreement (ChAFTA) and tariff rates are paid according to this agreement.
Customs Valuation
The dutiable value of an imported product is calculated by adding the cost, insurance, and freight (CIF) price.
Taxes
There are 3 main taxes payable: 1) Tariff duties 2) Value added Tax (VAT) 3) Consumption Tax – See next section to identify how much you need to pay for your product.
What is ChAFTA?
The China Australia Free Trade Agreement (ChAFTA) came into effect on 20 December 2015, significantly reducing the tariff barriers affecting bilateral trade in goods, including agricultural and F&B products. Under ChAFTA, the import duty on most processed food products from Australia will be zero from 1 January 2019.
How to assess my tariffs and taxes?
Step 1
Identify the product category and Check HS Code for your product.
Infant Formula includes formulated milk powder for infants, older infants and young children.
Infant Formula is classified as HS CODE 1901 and generally, under the sub-item of 1901.10.10.
There are 4 sub-items under 1901:
- 1901.10.10 Formula milk powder for infant use, put up for retail sale;
- 1901.10.90 Other food prepared for infant use, put up for retail sale;
- 1901.20.00 Mixes and doughs for the preparation of bakers’wares in 19.05;
- 1901.90.00 Other food preparations of malt extract, flour; dairy products.
English website for Import and Export Tariff Regulations and Import and Export Tariff (Goods and Item Note and Parsing): https://ftaportal.dfat.gov.au/CHN/AUS/ChAFTA/browse?selected=04015000
Step 2
Establish your Tariffs according to HS CODE.
The current ChAFTA tariff is 3% for this product. Prior to ChAFTA, the tariff for this product was 5%.
The relevant formula is:
Tariff = duty paid price × current applicable tariff rate.
Step 3
Establish your Consumption Tax according to HS CODE.
Most food don’t need to pay for Consumption Tax, except for wine at 10% and liquor at 20%.
So dairy products are not required to pay Consumption Tax.
Step 4
Establish your value-added taxes (VAT) according to HS CODE.
The VAT is 16% for HS CODE 1901.10.10.
The relevant formula is:
Import Value added tax = Import Value-added tax price × Import VAT Tax rate
Import VAT tax price = Customs Duty paid price + Customs duty + Import consumption tax
How to assess my tariffs and taxes?
Step 1
Identify the product category and Check HS Code for your product.
Infant Formula includes formulated milk powder for infants, older infants and young children.
Infant Formula is classified as HS CODE 1901 and generally, under the sub-item of 1901.10.10.
There are 4 sub-items under 1901:
- 1901.10.10 Formula milk powder for infant use, put up for retail sale;
- 1901.10.90 Other food prepared for infant use, put up for retail sale;
- 1901.20.00 Mixes and doughs for the preparation of bakers’wares in 19.05;
- 1901.90.00 Other food preparations of malt extract, flour; dairy products.
English website for Import and Export Tariff Regulations and Import and Export Tariff (Goods and Item Note and Parsing): https://ftaportal.dfat.gov.au/CHN/AUS/ChAFTA/browse?selected=04015000
Step 2
Establish your Tariffs according to HS CODE.
The current ChAFTA tariff is 3% for this product. Prior to ChAFTA, the tariff for this product was 5%.
The relevant formula is:
Tariff = duty paid price × current applicable tariff rate.
Step 3
Establish your Consumption Tax according to HS CODE.
Most food don’t need to pay for Consumption Tax, except for wine at 10% and liquor at 20%.
So dairy products are not required to pay Consumption Tax.
Step 4
Establish your value-added taxes (VAT) according to HS CODE.
The VAT is 16% for HS CODE 1901.10.10.
The relevant formula is:
Import Value added tax = Import Value-added tax price × Import VAT Tax rate
Import VAT tax price = Customs Duty paid price + Customs duty + Import consumption tax
How to assess my tariffs and taxes?
Step 1
Identify the product category and Check HS Code for your product.
Infant Formula includes formulated milk powder for infants, older infants and young children.
Infant Formula is classified as HS CODE 1901 and generally, under the sub-item of 1901.10.10.
There are 4 sub-items under 1901:
- 1901.10.10 Formula milk powder for infant use, put up for retail sale;
- 1901.10.90 Other food prepared for infant use, put up for retail sale;
- 1901.20.00 Mixes and doughs for the preparation of bakers’wares in 19.05;
- 1901.90.00 Other food preparations of malt extract, flour; dairy products.
English website for Import and Export Tariff Regulations and Import and Export Tariff (Goods and Item Note and Parsing): https://ftaportal.dfat.gov.au/CHN/AUS/ChAFTA/browse?selected=04015000
Step 2
Establish your Tariffs according to HS CODE.
The current ChAFTA tariff is 3% for this product. Prior to ChAFTA, the tariff for this product was 5%.
The relevant formula is:
Tariff = duty paid price × current applicable tariff rate.
Step 3
Establish your Consumption Tax according to HS CODE.
Most food don’t need to pay for Consumption Tax, except for wine at 10% and liquor at 20%.
So dairy products are not required to pay Consumption Tax.
Step 4
Establish your value-added taxes (VAT) according to HS CODE.
The VAT is 16% for HS CODE 1901.10.10.
The relevant formula is:
Import Value added tax = Import Value-added tax price × Import VAT Tax rate
Import VAT tax price = Customs Duty paid price + Customs duty + Import consumption tax
Traceability
What is dairy importation and sales record system?
According to “Administrative measures on inspection and quarantine of import and export dairy products” and “Administrative provisions on food importation and sales record”, food importers should establish a food importation and sales system.
Food importation record is a hard copy or electronic file that records imported food and relevant importing information. Food sales record is a hard copy or electronic file that records the vendors or consumers information. The inspection and quarantine organization will establish the credit record of dairy products importers.
Contents of the record system
The importation record system should include the following information:
Product name, brand, specification, quantity, manufacturing date or batch number, shelf life, country of origin, manufacturer name, name and contact information of exporters and vendors, delivery date, entry goods inspection and quarantine certificate number, etc.
The sales record system should include sales flow record and complaints-claims-recall record. All the records should be true and kept for at least 2 years.
Sales flow record should include:
Imported food name, specification, amount, weight, production batch number, sales date, buyer’s name and contact information, stock out number, Invoice number and recall process.
Complaints-claims-recall record should include:
Imported food name, specification, amount, weight, production date, production Lot No., complaints reasons, self-analysis, emergency handling and Follow-up improvement measures.
Cross Border E-commerce
Cross border e-commerce (CBEC) is a special import channel which allows goods to be sold online to consumers directly (B2C) via famous e-commerce platforms including: Alibaba’s Taobao, T-mall; JD.com, Kaola.com and others; opening the doors and market access to China’s e-commerce consumers. The CBEC channel has preferential policies on exemption for tariffs and regulatory requirements which usually applies to conventional general imports (B2B).
There are two models 1) CBEC Bonded Warehouse Model 2) CBEC Direct Mail Model, please see below for description:
1) CBEC bonded warehouse model
Products can be imported to any of the 15 approved CBEC bonded warehouse zones across China. Products are imported in bulk and temporarily stored in the bonded warehouse in the special pilot zone before an order is made by a consumer via a cross-border e-commerce website. After an e-sale order is made, products will be delivered from bonded area to the consumers directly via local courier. Please see pic 1 for visual flow diagram below:
2) CBEC Direct Mail Model
After the consumer places an order online, goods are shipped directly from overseas merchants (B2C) to consumer after customs clearance. Please see pic 2 for visual flow diagram below:
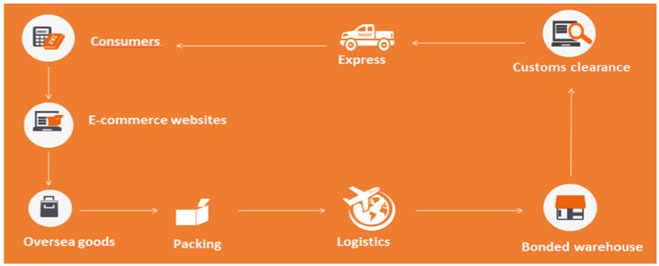
Only products listed in the positive list are allowed to be exported through Cross-border e-commerce
In April of 2016, China released two batches of positive list containing a list of 1,293 HS codes which can be imported /traded through CBEC, including packaged foods, UHT milk, beverages and others. Since the inception of the positive lists, all goods not included are banned on CBEC channel. Fortunately, most dairy products are included.
Is your product on positive lists?
Infant formula (HS code 19011010) is on the positive list. The article information of the HS code is as below.
| HS code | Article information |
| 19011010 | Powdered formulas prepared for infant use, put up for retail sale(Cocoa contents: <5% by weight of cocoa calculated on a totally defatted basis) |
Products on the positive lists are permitted to be imported via CBEC model which have preferential policies on regulatory requirements. Unlike general imports, infant formula exports do not need to comply with China labelling and product standards which are allowed to be imported until the end of 2018. But from 1 January 2019, all CBEC infant formula shall comply with China regulations. Below is the comparison of regulatory requirements between general import and CBEC model.
| Items | General Import | CBEC* | ||
| Comply with China GB standards | √ | X | ||
| Chinese labeling | √ | X | ||
| Tax (Tariff and VAT) | √ | √ | ||
| Pre-market Registration | √ | Before 1 Jan. 2019: X After 1 Jan. 2019: √ |
||
| Formulation compliance | √ | X | ||
* It is not sure whether regulatory requirements including GB standards, Chinese labelling and formulation compliance for CBEC will be the same as general import after 1 Jan. 2019.
CBEC Consumer Transaction Limits-RMB 2,000 per order & RMB 20,000 per year
For purchasing through CBEC, individual consumers are permitted to buy up to RMB 2,000 worth of such goods per order, up to a limit of RMB 20,000 per year.
CBEC Integrated Tax-11.2%
Products sold on CBEC are subject to 11.2% tax which is applied to the online retail price at the time of purchase (calculated as a 30% discount on VAT). However, if a single transaction exceeds the maximum value for single purchases or the combined annual total for each person, it will be taxed in full using the general trade tariff.
Suggestions for Exporters
Ensure regulatory compliance ASAP
Lack of consistency in regulations is one of the biggest challenges for companies engaging in CBEC. However, while CBEC remains an attractive channel to promote, market and sell products to Chinese buyers, Australian exporters are advised to ensure their products meet the China regulatory requirements & registration as early as possible before the end of the regulatory grace period on 31 December 2019.
Seek professional advice to track regulatory updates and ensure compliance.
What is professional overseas shoppers named “Daigou”?
Apart from CBEC, Daigou to Consumer (D2C) has been a popular choice with exporters to develop overseas brand awareness in the Chinese market with maximum flexibility and cost effectiveness.
A “Daigou” is an overseas personal shopper who purchases products for others in China and delivers it as personal postage. Usually, a Daigou earns a small margin/fee by charging the people buying the products. On 20 February 2018, Australia China Daigou Association was established aiming to facilitate the cooperation between Australia brands and Daigous and make more qualified Australia products enter the Chinese market.
Regulatory requirements for Daigou
Similar to CBEC, regulatory requirements for Daigou is much looser than general import. The comparison of regulatory requirements for infant formula between general import, CBEC and Daigou is as below.
| Regulatory requirements | General Import | CBEC | Daigou | |
| Comply with China GB standards | √ | X | X | |
| Chinese labeling | √ | X | X | |
| Tax (Tariff and VAT) | √ | √ | ?* | |
| Pre-market Registration | √ | Before 1 Jan. 2019: X After 1 Jan. 2019: √ |
X | |
| Formulation compliance | √ | X | X | |
* Daigou parcels are subject to tax only if customs inspected (the tax for foods is 15%, but can be exempted if tax value is less than 50RMB) , however due to significant volume of parcels flowing in and out of China, it is not possible for customs to check every parcel, leading to unclaimed taxes for many parcels via Daigou channel.
Suggestions: Daigou to test the market
Daigou has proven to be an effective stepping stone to test the market and to evaluate China consumer and cultural preferences to products; if utilised well, Daigou can be an effective marketing tool. However, sales via Daigou are considered by the authorities as being for use as “personal postage” for personal parcels. If operated at mass scale as a business, this may be considered as tax avoidance and smuggling on the extreme. It is suggested that, In the medium to long-term, the strategy should be to consider expansion and diversification to combine and consider a mix of CBEC and General Import channels which means registering products and complying with China standards to access general imports and retail stores (B2B).